细胞毒性双核η - 6-芳烃- ru (II)配合物:DNA序列特异性螺旋解绕和结合特性
IF 3.2
2区 化学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
采用核磁共振光谱和荧光滴定法,研究了分子式为{[(η - 6-cym)Ru(phe)]2(μ-BL)}Cl4的细胞毒性双功能DNA结合物与b型DNA序列d(5 ' -CGCGCG-3 ‘)、d(5 ’ -CTTTTGCAAAAG-3 ‘)和CT-DNA的结合性能,其中cym =对伞花烯,phe = 1,10-菲罗啉,BL = 4,4 ’ -联吡啶(BL-1)、(1)、1,2-二(4-吡啶基)乙烷(BL-2)、(2)和1,3-二(4-吡啶基)丙烷(BL-3)。结果显示,不同的结合模式和亲和性受DNA序列和BL长度的显著影响,相互作用是非选择性的,通过多种结合模式发生。复合物(1)被发现破坏w - c。C·G和A·T碱基对上的亚氨基氢键,有效地模拟了解旋酶对DNA链分离的机制。相反,(2)的结合不会破坏c - w。仅G·c序列d(5’-CTTTTGCAAAAG-3’)2中的GN1-H亚胺氢键可选择性地破坏富含at序列d(5’-CTTTTGCAAAAG-3’)中T2·A11碱基对的T2N3H亚胺氢键。配合物(3)诱导了d(5 ' -CGCGCG-3 ')2中心附近的螺旋局部解绕。复合物(1)-(3)的DNA结合亲和力表现出很强的序列依赖性,其结合常数(Kb)在1.23 × 103 M−1 ~ 6.34 × 105 M−1之间。本文章由计算机程序翻译,如有差异,请以英文原文为准。
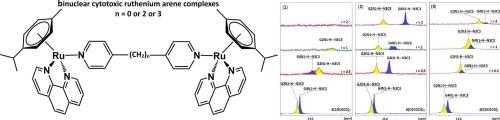
Cytotoxic binuclear η6-arene-Ru(II) complexes: DNA sequence specific helix unwinding and binding properties
Cytotoxic bifunctional DNA binders of the general formula {[(η6-cym)Ru(phe)]2(μ-BL)}Cl4, where cym = p-cymene, phe = 1,10-phenanthroline, BL = 4,4′-bipyridine (BL-1), (1), 1,2-bis(4-pyridyl)ethane (BL-2), (2) and 1,3-bis(4-pyridyl)propane (BL-3), (3), were investigated for their binding properties with B-type DNA sequences d(5′-CGCGCG-3′), d(5′-CTTTTGCAAAAG-3′) and CT-DNA using NMR spectroscopy and fluorescence titrations. The results revealed distinct binding modes and affinities, significantly influenced by both the DNA sequence and the length of the BL. The interactions were non-selective and occurred through multiple binding modes. Complex (1) was found to disrupt the W.-C. imino hydrogen bonds at both C·G and A·T base pairs, effectively mimicking the DNA strand separation mechanism by helicase enzymes. In contrast, the binding of (2) does not disrupt the C.-W. GN1-H imino hydrogen bonds in the G·C-only sequence d(5′-CGCGCG-3′)2 but it selectively disrupts the T2N3H imino hydrogen bond of the T2·A11 base pair in the AT-rich sequence d(5′-CTTTTGCAAAAG-3′). Complex (3) induced localized unwinding of the helix near the center of the d(5′-CGCGCG-3′)2. The DNA binding affinities of complexes (1)–(3) exhibit strong sequence dependence, with binding constants (Kb) ranging from 1.23 × 103 M−1 to 6.34 × 105 M−1.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Journal of Inorganic Biochemistry
生物-生化与分子生物学
CiteScore
7.00
自引率
10.30%
发文量
336
审稿时长
41 days
期刊介绍:
The Journal of Inorganic Biochemistry is an established international forum for research in all aspects of Biological Inorganic Chemistry. Original papers of a high scientific level are published in the form of Articles (full length papers), Short Communications, Focused Reviews and Bioinorganic Methods. Topics include: the chemistry, structure and function of metalloenzymes; the interaction of inorganic ions and molecules with proteins and nucleic acids; the synthesis and properties of coordination complexes of biological interest including both structural and functional model systems; the function of metal- containing systems in the regulation of gene expression; the role of metals in medicine; the application of spectroscopic methods to determine the structure of metallobiomolecules; the preparation and characterization of metal-based biomaterials; and related systems. The emphasis of the Journal is on the structure and mechanism of action of metallobiomolecules.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


