TTK 通过调节 ULK1 磷酸化和前核糖核酸剪接促进有丝分裂,从而抑制膀胱癌的线粒体凋亡
IF 15.4
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
膀胱癌(BC)仍然是一个主要的全球健康挑战,预后差,晚期治疗选择有限。TTK蛋白激酶(TTK)是一种丝氨酸/苏氨酸激酶,与多种癌症的进展有关,但其在BC中的作用尚未完全阐明。在这项研究中,我们发现TTK在BC组织和细胞系中显著上调,与患者预后不良相关。功能实验显示TTK能促进BC细胞增殖,抑制细胞凋亡。在机制上,TTK通过直接磷酸化ULK1的Ser477位点来增强有丝分裂,从而激活ULK1/ fundc1介导的有丝分裂途径。TTK敲低会破坏线粒体自噬,导致受损线粒体清除受损,线粒体活性氧(mtROS)过度积累,线粒体凋亡激活。此外,TTK磷酸化SRSF3的Ser108位点,阻止ULK1外显子5跳变并维持ULK1 mRNA的稳定性。这些发现表明TTK在维持BC细胞的有丝分裂中起关键作用。以TTK为靶点,通过破坏线粒体自噬和诱导线粒体凋亡,为治疗BC提供了新的途径。本文章由计算机程序翻译,如有差异,请以英文原文为准。
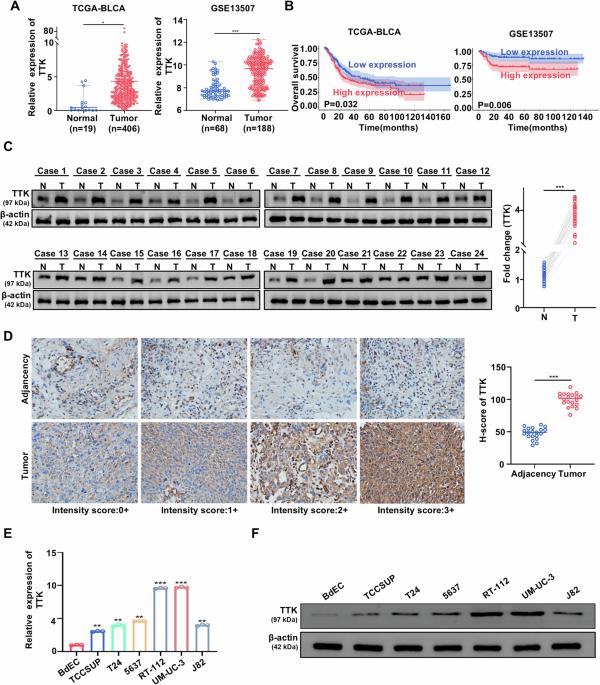

TTK promotes mitophagy by regulating ULK1 phosphorylation and pre-mRNA splicing to inhibit mitochondrial apoptosis in bladder cancer
Bladder cancer (BC) remains a major global health challenge, with poor prognosis and limited therapeutic options in advanced stages. TTK protein kinase (TTK), a serine/threonine kinase, has been implicated in the progression of various cancers, but its role in BC has not been fully elucidated. In this study, we show that TTK is significantly upregulated in BC tissues and cell lines, correlating with poor patient prognosis. Functional assays revealed that TTK promotes proliferation and inhibits apoptosis of BC cells. Mechanistically, TTK enhances mitophagy by directly phosphorylating ULK1 at Ser477, thereby activating the ULK1/FUNDC1-mediated mitophagy pathway. TTK knockdown disrupts mitophagy, leading to impaired clearance of damaged mitochondria, excessive accumulation of mitochondrial reactive oxygen species (mtROS), and activation of mitochondrial apoptosis. Furthermore, TTK phosphorylates SRSF3 at Ser108, preventing ULK1 exon 5 skipping and maintaining ULK1 mRNA stability. These findings show that TTK plays a key role in maintaining mitophagy in BC cells. Targeting TTK could offer a promising new approach for BC treatment by disrupting mitophagy and inducing mitochondrial apoptosis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell Death and Differentiation
生物-生化与分子生物学
CiteScore
24.70
自引率
1.60%
发文量
181
审稿时长
3 months
期刊介绍:
Mission, vision and values of Cell Death & Differentiation:
To devote itself to scientific excellence in the field of cell biology, molecular biology, and biochemistry of cell death and disease.
To provide a unified forum for scientists and clinical researchers
It is committed to the rapid publication of high quality original papers relating to these subjects, together with topical, usually solicited, reviews, meeting reports, editorial correspondence and occasional commentaries on controversial and scientifically informative issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


