CBX3通过CUL3/NRF2/GPX2轴抑制结直肠癌中的铁变态反应,从而促进多药耐受性
IF 7.3
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
化疗耐药是结直肠癌(CRC)治疗中的一个重大挑战。然而,化学耐药的机制尚不清楚。CBX3促进结直肠癌的增殖和转移。然而,CBX3在化疗耐药中的作用和机制尚不清楚。因此,我们旨在研究CBX3在结直肠癌多药耐药中的作用及其机制。我们的研究表明,较高水平的CBX3表达与较差的生存率相关,特别是在化疗后进展的组中。CBX3过表达增加伊立替康和奥沙利铂耐药性,而CBX3敲低抑制结直肠癌细胞的多药耐药。此外,CBX3抑制与多药耐药相关的铁下垂,并且铁下垂激活剂阻止CBX3过表达介导的细胞存活。RNA测序结果显示nrf2信号通路参与了这一过程。cbx3通过直接结合Cullin3启动子(CUL3)上调NRF2蛋白表达,抑制CUL3转录和CUL3介导的NRF2降解。此外,谷胱甘肽过氧化物酶2 (GPX2)在结直肠癌化疗耐药中处于CBX3-NRF2通路的下游。NRF2抑制剂ML385在PDX模型中抑制GPX2表达,增加铁下垂。我们的研究发现CBX3/NRF2/GPX2轴可能是介导结直肠癌多药耐药的新信号通路。这项研究提出了未来癌症治疗的新策略,以克服耐药性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
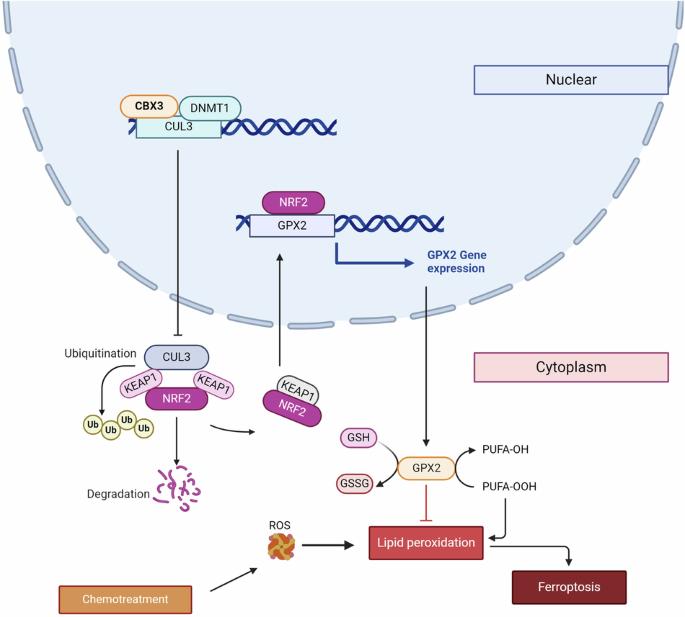
CBX3 promotes multidrug resistance by suppressing ferroptosis in colorectal carcinoma via the CUL3/NRF2/GPX2 axis
Chemoresistance poses a significant challenge in colorectal cancer (CRC) treatment. However, the mechanisms underlying chemoresistance remain unclear. CBX3 promoted proliferation and metastasis in CRC. However, the role and mechanism of CBX3 in chemoresistance remain unknown. Therefore, we aimed to investigate the effects and mechanisms of CBX3 on multidrug resistance in CRC. Our studies showed that higher levels of CBX3 expression were associated with poor survival, especially in groups with progression following chemotherapy. CBX3 overexpression increased Irinotecan and Oxaliplatin resistance, whereas CBX3 knockdown suppressed multidrug resistance in CRC cells. Additionally, CBX3 inhibited ferroptosis associated with multidrug resistance, and the ferroptosis activators prevented CBX3 overexpression-mediated cell survival. RNA sequencing revealed that the NRF2-signaling pathway was involved in this process. CBX3-upregulated NRF2 protein expression by directly binding to the promoter of Cullin3 (CUL3) to suppress CUL3 transcription and CUL3-mediated NRF2 degradation. Moreover, Glutathione Peroxidase 2 (GPX2) was downstream of the CBX3-NRF2 pathway in CRC chemoresistance. ML385, an NRF2 inhibitor, suppressed GPX2 expression, and increased ferroptosis in PDX models. Our study identified CBX3/NRF2/GPX2 axis may be a novel signaling pathway that mediates multidrug resistance in CRC. This study proposes developing novel strategies for cancer treatment to overcome drug resistance in the future.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Oncogene
医学-生化与分子生物学
CiteScore
15.30
自引率
1.20%
发文量
404
审稿时长
1 months
期刊介绍:
Oncogene is dedicated to advancing our understanding of cancer processes through the publication of exceptional research. The journal seeks to disseminate work that challenges conventional theories and contributes to establishing new paradigms in the etio-pathogenesis, diagnosis, treatment, or prevention of cancers. Emphasis is placed on research shedding light on processes driving metastatic spread and providing crucial insights into cancer biology beyond existing knowledge.
Areas covered include the cellular and molecular biology of cancer, resistance to cancer therapies, and the development of improved approaches to enhance survival. Oncogene spans the spectrum of cancer biology, from fundamental and theoretical work to translational, applied, and clinical research, including early and late Phase clinical trials, particularly those with biologic and translational endpoints.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



