钯催化二烯和三烯的位点选择性区域发散碳环化:取代环己烯和环丁烯之间的切换
IF 15.6
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
自然界通过在二烯或多烯的特定烯烃位点上的环化反应,有效地从常见的线性前体产生无数结构多样的碳环框架。相比之下,实现二烯或多烯底物的位点选择性功能化仍然是化学合成中的一个巨大挑战。在此,我们报道了一对高位点选择性,区域发散的二烯烯和三烯烯的碳环化反应,使得从具有高非对映选择性的共同前体高效合成顺式-1,4-二取代环己烯和反式-1,2-二取代环丁烯。值得注意的是,简单的非手性有机酸和胺被确定为控制这些钯催化的区域发散碳环化的强大配体。该方法代表了第一个位置选择性区域发散碳环化的例子,为热动力学上不利的顺式-1,4-二取代环己烯和完全取代的反式-1,2-环丁烯的立体定向合成提供了一种实用的方法。此外,所开发的方法为金属催化的二烯和多烯前体的位点选择性、区域发散性碳环化的发展提供了一般见解,模拟了自然碳环化过程。本文章由计算机程序翻译,如有差异,请以英文原文为准。
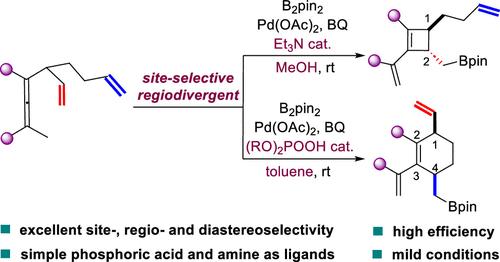
Palladium-Catalyzed Site-Selective Regiodivergent Carbocyclization of Di- and Trienallenes: A Switch between Substituted Cyclohexene and Cyclobutene
Nature efficiently produces a myriad of structurally diverse carbon ring frameworks from common linear precursors via cyclization reactions at specific olefinic sites in dienes or polyenes. In contrast, achieving the site-selective functionalization of diene or polyene substrates remains a formidable challenge in chemical synthesis. Herein, we report a pair of highly site-selective, regiodivergent carbocyclization reactions of dienallenes and trienallenes, enabling the efficient synthesis of cis-1,4-disubstituted cyclohexenes and trans-1,2-disubstituted cyclobutenes from a common precursor with high diastereoselectivity. Remarkably, simple achiral organophosphoric acids and amines are identified as powerful ligands for controlling these palladium-catalyzed regiodivergent carbocyclizations. This approach represents the first example of site-selective regiodivergent carbocyclization, providing a practical method for the stereospecific synthesis of thermodynamically disfavored cis-1,4-disubstituted cyclohexenes and fully substituted trans-1,2-cyclobutenes. Additionally, the methodology developed offers general insights into the development of metal-catalyzed site-selective, regiodivergent carbocyclizations of diene and polyene precursors, mimicking natural carbocyclization processes.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



