慢性淋巴细胞白血病肿瘤诱导免疫功能紊乱的基础上的胆固醇稳态和脂质筏动力学。
IF 19.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
自体t细胞疗法对慢性淋巴细胞白血病(CLL)的疗效有限,其中获得性免疫功能障碍普遍存在。在CLL中,线粒体代谢紊乱与t细胞活化和增殖缺陷有关。最近的研究表明,脂质代谢调节线粒体功能和T细胞的分化,但其在CLL中的作用仍未被探索。这项综合研究比较了CLL患者和健康供者的t细胞脂质代谢,揭示了tcr介导激活后人类t细胞扩增对外源性胆固醇的严重依赖。通过多组学和功能分析,我们发现CLL患者存活冷冻样本中的T细胞(CLL T细胞)对胆固醇剥夺的适应受损,关键脂质代谢转录因子上调不足。CLL T细胞表现出脂质储存改变,甘油三酯增加,胆固醇降低,脂肪酸氧化效率低下(FAO)。使用来自固有FAO紊乱患者的样本,研究了T细胞中FAO减少的功能后果。降低的FAO与较低的t细胞活化有关,但不影响增殖。这意味着低胆固醇水平是限制CLL中t细胞增殖的主要因素。CLL T细胞显示出越来越少的聚集性脂筏,这可能解释了在这些患者中观察到的免疫突触形成受损。我们的研究结果强调了脂质代谢的显著破坏是CLL T细胞功能缺陷的驱动因素,强调了胆固醇在T细胞增殖中的关键作用。这项研究表明,调节胆固醇代谢可以增强CLL中的t细胞功能,提出了新的免疫治疗方法来改善这种具有挑战性的疾病的预后。本文章由计算机程序翻译,如有差异,请以英文原文为准。
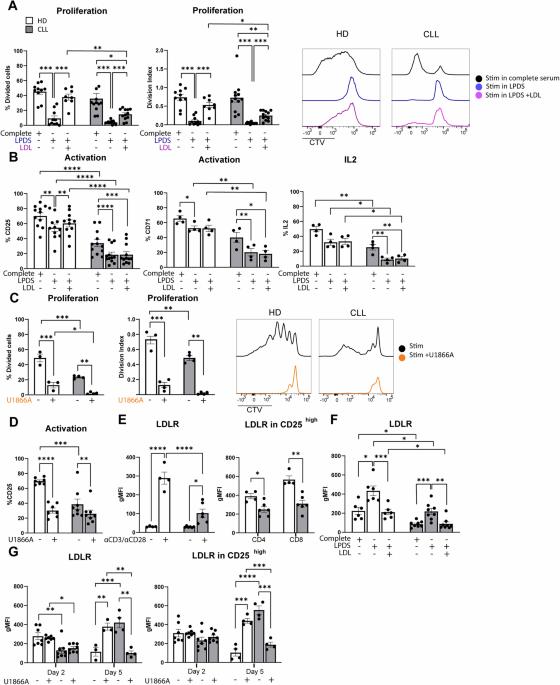
Cholesterol homeostasis and lipid raft dynamics at the basis of tumor-induced immune dysfunction in chronic lymphocytic leukemia
Autologous T-cell therapies show limited efficacy in chronic lymphocytic leukemia (CLL), where acquired immune dysfunction prevails. In CLL, disturbed mitochondrial metabolism has been linked to defective T-cell activation and proliferation. Recent research suggests that lipid metabolism regulates mitochondrial function and differentiation in T cells, yet its role in CLL remains unexplored. This comprehensive study compares T-cell lipid metabolism in CLL patients and healthy donors, revealing critical dependence on exogenous cholesterol for human T-cell expansion following TCR-mediated activation. Using multi-omics and functional assays, we found that T cells present in viably frozen samples of patients with CLL (CLL T cells) showed impaired adaptation to cholesterol deprivation and inadequate upregulation of key lipid metabolism transcription factors. CLL T cells exhibited altered lipid storage, with increased triacylglycerols and decreased cholesterol, and inefficient fatty acid oxidation (FAO). Functional consequences of reduced FAO in T cells were studied using samples from patients with inherent FAO disorders. Reduced FAO was associated with lower T-cell activation but did not affect proliferation. This implicates low cholesterol levels as a primary factor limiting T-cell proliferation in CLL. CLL T cells displayed fewer and less clustered lipid rafts, potentially explaining the impaired immune synapse formation observed in these patients. Our findings highlight significant disruptions in lipid metabolism as drivers of functional deficiencies in CLL T cells, underscoring the pivotal role of cholesterol in T-cell proliferation. This study suggests that modulating cholesterol metabolism could enhance T-cell function in CLL, presenting novel immunotherapeutic approaches to improve outcome in this challenging disease.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


