单独的含氧海马体通过增强情境记忆和动机来塑造饮食选择
IF 18.9
1区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
摘要
海马体(HPC)已经成为控制食物摄入的关键角色,超出了它在记忆中的众所周知的作用。虽然先前的研究主要将HPC与食物摄入抑制联系起来,但最近的研究表明HPC在食欲过程中起作用。在这里,我们确定了背侧HPC (dHPC)内空间上不同的神经元群,它们对脂肪或糖做出反应,这是促进肥胖发展的有效天然强化物。利用活动依赖的营养反应dHPC神经元的遗传捕获,我们证明了这两个群体通过不同的机制促进营养特异性摄入的因果作用。糖反应神经元编码糖位置的空间记忆,而脂肪反应神经元选择性地增强对脂肪摄入的偏好和动机。重要的是,刺激任何一种营养反应的dHPC神经元都会增加食物摄入,而消融对致肥性饮食消耗的影响不同,并阻止饮食引起的体重增加。总的来说,这些发现揭示了以前未知的巨量营养素特异性消耗背后的摄氧回路,并为开发潜在的肥胖治疗方法提供了基础。本文章由计算机程序翻译,如有差异,请以英文原文为准。

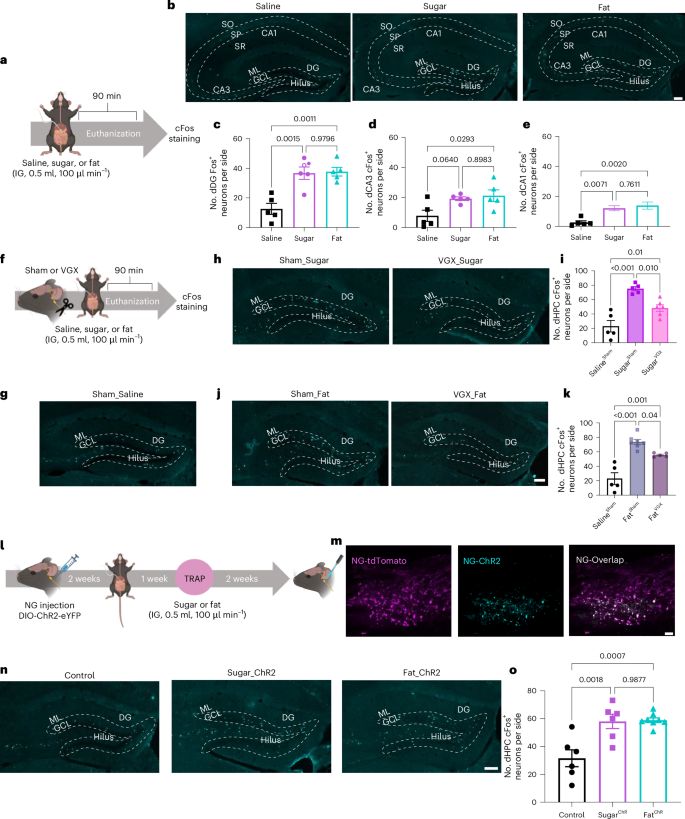
Separate orexigenic hippocampal ensembles shape dietary choice by enhancing contextual memory and motivation
The hippocampus (HPC) has emerged as a critical player in the control of food intake, beyond its well-known role in memory. While previous studies have primarily associated the HPC with food intake inhibition, recent research suggests a role in appetitive processes. Here we identified spatially distinct neuronal populations within the dorsal HPC (dHPC) that respond to either fats or sugars, potent natural reinforcers that contribute to obesity development. Using activity-dependent genetic capture of nutrient-responsive dHPC neurons, we demonstrate a causal role of both populations in promoting nutrient-specific intake through different mechanisms. Sugar-responsive neurons encoded spatial memory for sugar location, whereas fat-responsive neurons selectively enhanced the preference and motivation for fat intake. Importantly, stimulation of either nutrient-responsive dHPC neurons increased food intake, while ablation differentially impacted obesogenic diet consumption and prevented diet-induced weight gain. Collectively, these findings uncover previously unknown orexigenic circuits underlying macronutrient-specific consumption and provide a foundation for developing potential obesity treatments. Yang et al. identify and characterize distinct sugar- and fat-responsive neuronal populations in the dorsal hippocampus and their contribution to the development of obesity.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature metabolism
ENDOCRINOLOGY & METABOLISM-
CiteScore
27.50
自引率
2.40%
发文量
170
期刊介绍:
Nature Metabolism is a peer-reviewed scientific journal that covers a broad range of topics in metabolism research. It aims to advance the understanding of metabolic and homeostatic processes at a cellular and physiological level. The journal publishes research from various fields, including fundamental cell biology, basic biomedical and translational research, and integrative physiology. It focuses on how cellular metabolism affects cellular function, the physiology and homeostasis of organs and tissues, and the regulation of organismal energy homeostasis. It also investigates the molecular pathophysiology of metabolic diseases such as diabetes and obesity, as well as their treatment. Nature Metabolism follows the standards of other Nature-branded journals, with a dedicated team of professional editors, rigorous peer-review process, high standards of copy-editing and production, swift publication, and editorial independence. The journal has a high impact factor, has a certain influence in the international area, and is deeply concerned and cited by the majority of scholars.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


