重氮嘧啶作为瓜脲类大环的客体:光响应结合和热异构化的超分子催化
IF 15.6
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
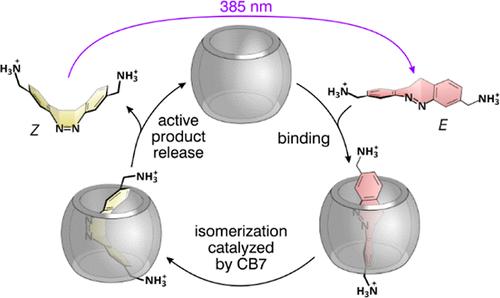
超分子主客体复合物的光开关是许多分子控制宏观功能的基础,如溶胶-凝胶转变、光药理学、离子或分子的主动运输、光动力分子机器等等。最常用的系统采用光活性偶氮苯客体和合成宿主分子,它们结合为稳定的E异构体,并在暴露于紫外线后作为Z形式解离。我们提出了一种新的,非常高效的葫芦[7]uril (CB7)/重氮嘧啶主/客体配合物,具有倒置稳定性,在紫外线照射下自组装,在黑暗中解离。Z和E异构体在水中的缔合常数相差超过104倍。我们还发现,CB7显著加速了热激活的E→Z异构化,这是一种罕见的通过过渡态稳定而没有产物抑制的类酶催化。与CB7相比,葫芦[8]uril (CB8)与两种异构体具有高亲和力,对Z异构体具有良好的选择性(约1000倍)。值得注意的是,相对于CB7,该异构体优先结合CB8的因子大于1 × 106。我们还使用该系统引入了一种超分子光酸,该光酸建立在与CB7结合的客体的碱性增加和E异构体的极高亲和力的基础上,该异构体被用来取代CB7中的酸,从而改变溶液的pH值。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Diazocines as Guests of Cucurbituril Macrocycles: Light-Responsive Binding and Supramolecular Catalysis of Thermal Isomerization
The photoswitching of supramolecular host–guest complexes is the basis of numerous molecularly controlled macroscopic functions, such as sol–gel transition, photopharmacology, the active transport of ions or molecules, light-powered molecular machines, and much more. The most commonly used systems employ photoactive azobenzene guests and synthetic host molecules, which bind as the stable E isomers and dissociate as the Z forms after exposure to UV light. We present a new, extraordinarily efficient cucurbit[7]uril (CB7)/diazocine host/guest complex with inverted stability that self-assembles under UV irradiation and dissociates in the dark. The association constants of the Z and E isomers in water differ by more than 104-fold. We also show that the thermally activated E → Z isomerization is significantly accelerated by CB7, which is a rare case of enzyme-like catalysis by transition state stabilization without product inhibition. In contrast to CB7, cucurbit[8]uril (CB8) binds both isomers with high affinity, showing good selectivity (∼1000-fold) toward the Z isomer. Notably, this isomer preferentially binds CB8 relative to CB7 by a factor greater than 1 × 106. We also use the system to introduce a supramolecular photoacid that builds on the increased basicity of a guest bound to CB7 and on the extremely high affinity of the E isomer, which is utilized to displace the acid from CB7, thereby switching the pH of the solution.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


