铁催化芳酰氯和三氟酸盐的宫宫硼化反应
IF 5
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
简单芳基氯化物代表具有挑战性的底物在铁催化硼化。Li[B(tBu)pin-Bpin]作为硼化试剂和由三酸铁(II)和市售n-杂环碳烯配体(IMes)原位形成的催化剂的组合,比以前报道的铁基催化剂具有显著提高的活性和更广泛的应用范围。三氟化铁也是三氟化芳基硼化的良好预催化剂──这是一种以前未报道过的转化──在这种情况下,不需要使用times配体。本文章由计算机程序翻译,如有差异,请以英文原文为准。
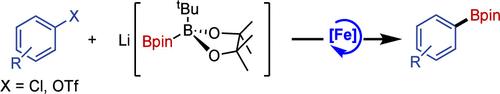
Iron-Catalyzed Miyaura Borylation of Aryl Chlorides and Triflates
Simple aryl chlorides represent challenging substrates in iron-catalyzed borylation. A combination of Li[B(tBu)pin-Bpin] as the borylating reagent and a catalyst formed in situ from iron(II) triflate and the commercially available N-heterocyclic carbene ligand, IMes, gives significantly improved activity and a much broader scope than previously reported iron-based catalysts. Iron triflate is also a good precatalyst for the borylation of aryl triflates─a previously unreported transformation─and in these cases the IMes ligand is not required.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



