多组学分析结合网络药理学揭示鲁泰卡品治疗慢性萎缩性胃炎的机制
IF 5.4
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
民族药理学意义芸香四逆散(A.Juss. T.G.Hartley)是一种具有数千年历史的传统中药,在缓解胃痛、消化不良、呕吐和腹泻方面发挥着重要作用。芦根碱(RUT)是 Tetradium ruticarpum (A.Juss.) T.G.Hartley 的主要活性成分之一,对慢性萎缩性胃炎(CAG)具有潜在的治疗活性。材料和方法用 0.1% 氨水和 20 mmol/L 脱氧胆酸钠溶液诱导 SD 大鼠建立 CAG 模型,同时采用不规则禁食周期。通过病理检查和血清生化指标(包括 PP、IL-6、MTL、TNF-α、PG I、SS、PG II、IL-10 和 GAS-17)评估 RUT 治疗 CAG 的疗效。随后,进行了网络药理学、16s rRNA 测序、转录组学和广泛的靶向代谢组学研究,以揭示 RUT 治疗 CAG 的作用机制。结果RUT显著改善了CAG大鼠的体重、胃液pH值和胃组织损伤。血清生化指标结果显示,RUT 能明显抑制 SS、GAS-17、IL-6 和 TNF-α 的表达水平,提高 MTL、PP、PGI、PGII 和 IL-10 的水平。此外,RUT 还明显增加了粘膜屏障蛋白的表达,如 ZO-1、E-cadherin、claudin-4 和 occludin。网络药理学结合转录组学发现,MAPK 信号通路是 RUT 治疗 CAG 的最重要通路。进一步分析表明,RUT通过调节亚油酸代谢、代谢通路等主要与能量代谢相关的途径,有效改善了CAG大鼠的胃组织代谢紊乱。基于 16S rRNA 基因的微生物区系分析表明,RUT 改变了肠道微生物区系的组成,降低了未分类的毛里杆菌科微生物的相对丰度。PICRUST 分析表明,差异菌可能参与了能量代谢途径的调节,从而改善了 CAG。对转录组和代谢组的综合分析表明,RUT 通过调控 TGER2、CBR1 和 CTPS1 靶点改善了不同代谢产物。这为 RUT 如何对 CAG 发挥治疗作用提供了新的见解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
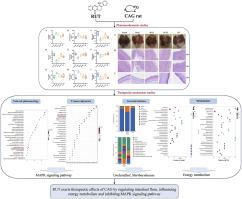
Multi-omics analysis combined with network pharmacology revealed the mechanisms of rutaecarpine in chronic atrophic gastritis
Ethnopharmacological relevance
Tetradium ruticarpum (A.Juss.) T.G.Hartley is a traditional Chinese medicine with a history of thousands of years, which plays an important role in the relief of gastric pain, indigestion, vomiting and diarrhea. Rutaecarpine (RUT) is one of the major active constituents of Tetradium ruticarpum (A.Juss.) T.G.Hartley with potential therapeutic activity in chronic atrophic gastritis (CAG). However, the mechanism of RUT to improve CAG is not well understood.
Aim of this study
This study aimed to evaluate the efficacy of RUT in treating CAG and its underlying mechanism.
Materials and methods
The CAG model of SD rats was established by induction with 0.1% ammonia and 20 mmol/L sodium deoxycholate solution, accompanied with irregular fasting cycle. The efficacy of RUT in treating CAG was assessed through pathological examination and serum biochemical indices including PP, IL-6, MTL, TNF-α, PG I, SS, PG II, IL-10 and GAS-17. Following this, network pharmacology, 16s rRNA sequencing, transcriptomics, and broadly targeted metabolomics were conducted to unravel the underlying mechanisms of RUT's action in CAG treatment. Ultimately, molecular docking, western blotting, and immunohistochemistry were employed to validate the critical targets and pathways involved in RUT's therapeutic approach for CAG.
Results
RUT significantly improved body weight, gastric juice pH and gastric histologic injury in CAG rats. The results of serum biochemical indices showed that RUT significantly inhibited the expression levels of SS, GAS-17, IL-6 and TNF-α, and increased the levels of MTL, PP, PGI, PGII and IL-10. In addition, RUT apparently increased the expression of mucosal barrier proteins such as ZO-1, E-cadherin and claudin-4 and occludin. Network pharmacology in combination with transcriptomics revealed that the MAPK signaling pathway was the most important pathway for RUT treatment of CAG. Further analysis suggested that by regulating linoleic acid metabolism, metabolic pathways, etc. mainly related to energy metabolism, RUT intervention effectively ameliorated gastric tissue metabolic disorders in CAG rats. The 16S rRNA gene-based microbiota analysis revealed that RUT altered the composition of the intestinal microbiota and decreased the relative abundance of unclassified_Muribaculaceae. PICRUST analysis suggested that the differential bacteria may be involved in energy metabolism pathway regulation for the improvement of CAG. A comprehensive analysis of the transcriptome and metabolome showed that the RUT improved the differential metabolites through the regulation of TGER2, CBR1 and CTPS1 targets.
Conclusion
These findings indicated that RUT's mechanism of action in treating CAG was related to modulating the gut microbiota, influencing energy metabolism, and inhibiting the MAPK signaling pathway. This provided new insights into how RUT exerts its therapeutic effects on CAG.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of ethnopharmacology
医学-全科医学与补充医学
CiteScore
10.30
自引率
5.60%
发文量
967
审稿时长
77 days
期刊介绍:
The Journal of Ethnopharmacology is dedicated to the exchange of information and understandings about people''s use of plants, fungi, animals, microorganisms and minerals and their biological and pharmacological effects based on the principles established through international conventions. Early people confronted with illness and disease, discovered a wealth of useful therapeutic agents in the plant and animal kingdoms. The empirical knowledge of these medicinal substances and their toxic potential was passed on by oral tradition and sometimes recorded in herbals and other texts on materia medica. Many valuable drugs of today (e.g., atropine, ephedrine, tubocurarine, digoxin, reserpine) came into use through the study of indigenous remedies. Chemists continue to use plant-derived drugs (e.g., morphine, taxol, physostigmine, quinidine, emetine) as prototypes in their attempts to develop more effective and less toxic medicinals.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


