基于噻唑烷二酮的麦角甾醇过氧化物结构修饰提供了噻唑烷二酮共轭衍生物,这些衍生物可通过 PI3K/AKT/mTOR 途径对乳腺癌细胞产生强效作用。
IF 3.3
3区 医学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
过氧化麦角甾醇(EP)是从传统中药灵芝中分离出来的一种甾体化合物。然而,EP 受限于其溶解性和在抗肿瘤研究中的中等效力。本研究设计并合成了一系列新型过氧化麦角甾醇-3-噻唑烷二酮衍生物,通过改变过氧化麦角甾醇与噻唑烷二酮之间的连接物,有望获得抗肿瘤活性更强的化合物。细胞毒性筛选结果表明,化合物 13o 是对 MCF-7 细胞株最有效的衍生物,其 IC50 值为 3.06 μM,与母体 EP 相比,具有更强的抗肿瘤活性。进一步的体外和体内研究表明,化合物 13o 可通过抑制 PI3K/Akt/mTOR 通路,降低线粒体膜电位,增加活性氧水平,阻滞 G0/G1 期细胞周期,诱导肿瘤细胞凋亡。体内 4T1 乳腺癌小鼠模型显示,13o 不仅能持续抑制肿瘤增殖,而且效果强于市售药物 5-氟尿嘧啶。研究结果表明,化合物 13o 可能是开发新型乳腺癌化疗药物的一种新型、高效、低毒的结构先导化合物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
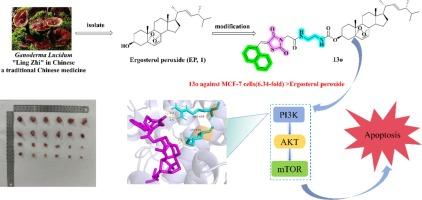
Thiazolidinedione-based structure modification of ergosterol peroxide provides thiazolidinedione-conjugated derivatives as potent agents against breast cancer cells through a PI3K/AKT/mTOR pathway
Ergosterol peroxide (EP) is a steroidal compound isolated from the traditional Chinese medicine Ganoderma lucidum. However, EP is limited by its solubility and moderate potency in antitumor studies. In the present study, a series of novel ergosterol peroxide-3-thiazolidinedione derivatives were designed and synthesized, by changing the linker between ergosterol peroxide and thiazolidinedione, it is expected to obtain compounds with better antitumor activity. The cytotoxicity screening showed that compound 13o is the most active derivative against the MCF-7 cell line with an IC50 of 3.06 μM, and exhibited stronger antitumor activity compared to the parent EP. Further in vitro and vivo studies showed that compound 13o may reduced the mitochondrial membrane potential, increased the reactive oxygen species level and blocked the cell cycle in G0/G1 phase, and induced apoptosis of tumor cells by inhibiting the PI3K/Akt/mTOR pathway. In vivo 4T1 mouse model of breast cancer showed that 13o not only continued to inhibit tumor proliferation but also had a stronger effect than the marketed drug 5-fluorouracil, compound 13o had a good safety profile in vivo. The results suggest that compound 13o may represent a novel, highly potent and low-toxicity structural lead for the development of new breast cancer chemotherapies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Bioorganic & Medicinal Chemistry
医学-生化与分子生物学
CiteScore
6.80
自引率
2.90%
发文量
413
审稿时长
17 days
期刊介绍:
Bioorganic & Medicinal Chemistry provides an international forum for the publication of full original research papers and critical reviews on molecular interactions in key biological targets such as receptors, channels, enzymes, nucleotides, lipids and saccharides.
The aim of the journal is to promote a better understanding at the molecular level of life processes, and living organisms, as well as the interaction of these with chemical agents. A special feature will be that colour illustrations will be reproduced at no charge to the author, provided that the Editor agrees that colour is essential to the information content of the illustration in question.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


