肠道微生物群衍生的 3-吲哚乙酸通过小胶质细胞芳基烃受体对败血症相关脑病起到保护作用
IF 4.6
2区 医学
Q1 NEUROSCIENCES
引用次数: 0
摘要
背景:肠道微生物群对中枢神经系统疾病的发病机制有重要影响。在肠道微生物群产生的生物活性分子中,3-吲哚乙酸(IAA)已被证明可减轻氧化应激和炎症反应。本实验旨在确定IAA对败血症相关脑病(SAE)的影响及其内在机制:方法:共有 34 名脓毒症患者和 24 名健康对照者参与了粪便 IAA 与脓毒症脑病之间临床相关性的分析。粪便微生物群移植用于验证肠道微生物群及其代谢产物在脓毒症脑病中的作用。年龄在六到八周的雄性 C57BL/6 小鼠经口服 IAA 预处理后,接受盲肠结扎和穿刺(CLP)手术。该疗法与芳基烃受体(AhR)拮抗剂 CH223191 或 CSF1R 抑制剂 PLX3397 联合使用,以消除小胶质细胞。免疫荧光染色法和酶联免疫吸附试验被用来评估小胶质细胞的活化和炎性细胞因子的分泌。行为评估用于量化神经功能缺陷:结果:脓毒症相关谵妄(SAD)患者粪便中的IAA水平降低,这是SAE的一种表现形式。IAA水平的降低与临床预后的恶化密切相关。移植SAD患者的粪便微生物群可诱导小鼠出现SAE样表型,但补充外源IAA可改善由小胶质细胞介导的SAE样表型。IAA能有效地与芳基烃受体(AhR)结合。此外,IAA还能提高经脂多糖(LPS)处理的小胶质细胞中AhR的核活性,从而减少炎性细胞因子的分泌。AhR抑制剂CH223191抵消了IAA对小鼠SAE的保护作用:结论:肠道微生物群衍生的 IAA 可通过激活小胶质细胞中的 AhR 来保护小鼠免受 SAE 的伤害,从而改善神经元和认知障碍。因此,IAA有望成为控制SAE的潜在治疗药物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
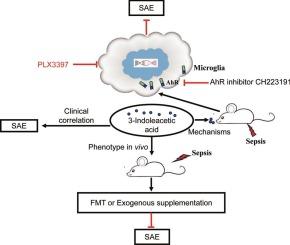
Gut microbiota-derived 3-indoleacetic acid confers a protection against sepsis-associated encephalopathy through microglial aryl hydrocarbon receptors
Background
The gut microbiota significantly contributes to the pathogenesis of central nervous system disorders. Among the bioactive molecules produced by the gut microbiota, 3-indoleacetic acid (IAA) has been shown to attenuate oxidative stress and inflammatory responses. This experiment aimed to determine the impacts of IAA on sepsis-associated encephalopathy (SAE) and the underlying mechanisms.
Methods
A total of 34 septic patients and 24 healthy controls were included in the analysis of the clinical correlation between fecal IAA and septic encephalopathy. Fecal microbiota transplantation was used to verify the role of the gut microbiota and its metabolites in SAE. Male C57BL/6 mice aged six to eight weeks, pre-treated with IAA via oral gavage, were subjected to the cecal ligation and puncture (CLP) procedures. This treatment was administered either in combination with an aryl hydrocarbon receptor (AhR) antagonist, CH223191, or a CSF1R inhibitor, PLX3397, to eliminate microglia. Both immunofluorescence staining and enzyme-linked immunosorbent assays were used to evaluate microglia activation and inflammatory cytokine secretion. Behavioral assessments were conducted to quantify neurological deficits.
Results
A decreased fecal level of IAA was observed in the patients with sepsis-associated delirium (SAD), a manifestation of SAE. A reduced IAA level was significantly associated with worsen clinical outcomes. Fecal microbiota transplantation from the SAD patients induced an SAE-like phenotype in mice, but supplementing exogenous IAA improved the SAE-like phenotype, mediated by microglia. IAA effectively binded with the aryl hydrocarbon receptor (AhR). Furthermore, IAA increased the nuclear activity of AhR in the lipopolysaccharide (LPS)-treated microglial cells, leading to reduced secretion of inflammatory cytokines. The AhR inhibitor CH223191 counteracted the protective effect of IAA against SAE in mice.
Conclusions
Gut microbiota-derived IAA confers a protection against SAE by activating AhR in microglia, improving neuronal and cognitive impairments. Thus, IAA holds the promise as a potential therapeutic agent for managing SAE.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Experimental Neurology
医学-神经科学
CiteScore
10.10
自引率
3.80%
发文量
258
审稿时长
42 days
期刊介绍:
Experimental Neurology, a Journal of Neuroscience Research, publishes original research in neuroscience with a particular emphasis on novel findings in neural development, regeneration, plasticity and transplantation. The journal has focused on research concerning basic mechanisms underlying neurological disorders.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


