靶向 TAMs:我们能比癌细胞更聪明吗?
IF 21.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
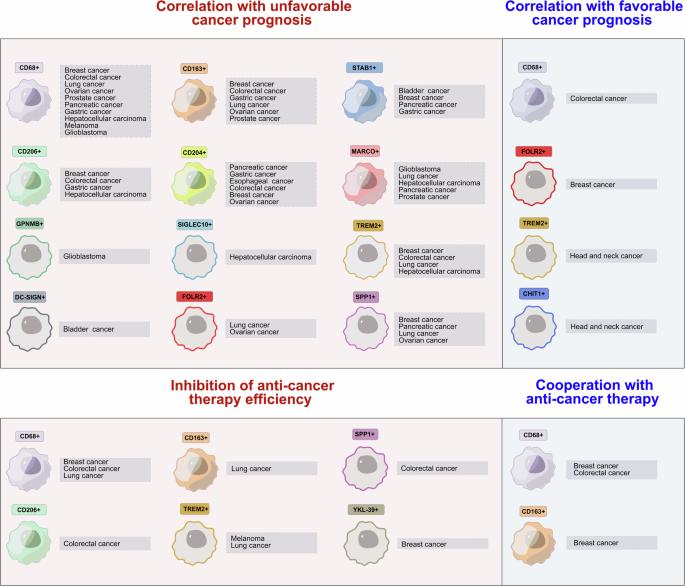
摘要:随着癌症发病率和发病地域的不断扩大,癌症已成为全球死亡、生活质量下降和残疾的主要原因之一。在开发新的抗癌疗法、提高免疫治疗工具的效率以及将传统疗法人性化方面取得的主要进展需要考虑癌症特异性和患者特异性的先天免疫程序。瘤内 TAMs 及其前体、常驻巨噬细胞和单核细胞是肿瘤进展和耐药性的主要调节因素。我们的综述总结了有关 TAMs 亚群及其越来越多的生物标志物的累积证据,指出了它们对癌变和耐药性临床参数的预测价值,重点是非感染性病因的实体瘤。我们介绍了有关 TAMs 在肿瘤进展各个阶段的肿瘤支持功能的最新知识,并重点介绍了最近通过单细胞和空间分析方法发现的生物标志物,这些生物标志物可区分肿瘤促进型和肿瘤抑制型 TAMs,其中这两种亚型均表达原型 M1 和 M2 基因的组合。我们的综述侧重于 TAMs 中表观遗传、信号传导、转录和代谢途径之间相互影响的新机制。我们特别关注了最近发现的癌细胞新陈代谢与组蛋白乳酰化对 TAMs 的表观遗传学编程之间的联系,这可能是 TAMs 无限原瘤编程的原因。最后,我们解释了 TAM 如何干扰目前使用的抗癌疗法,并总结了临床试验的最先进数据,我们将其分为四类:抑制 TAM 的存活和分化、抑制单核细胞/TAM 招募进入肿瘤、TAM 的功能重编程以及巨噬细胞的遗传增强。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Targeting of TAMs: can we be more clever than cancer cells?
With increasing incidence and geography, cancer is one of the leading causes of death, reduced quality of life and disability worldwide. Principal progress in the development of new anticancer therapies, in improving the efficiency of immunotherapeutic tools, and in the personification of conventional therapies needs to consider cancer-specific and patient-specific programming of innate immunity. Intratumoral TAMs and their precursors, resident macrophages and monocytes, are principal regulators of tumor progression and therapy resistance. Our review summarizes the accumulated evidence for the subpopulations of TAMs and their increasing number of biomarkers, indicating their predictive value for the clinical parameters of carcinogenesis and therapy resistance, with a focus on solid cancers of non-infectious etiology. We present the state-of-the-art knowledge about the tumor-supporting functions of TAMs at all stages of tumor progression and highlight biomarkers, recently identified by single-cell and spatial analytical methods, that discriminate between tumor-promoting and tumor-inhibiting TAMs, where both subtypes express a combination of prototype M1 and M2 genes. Our review focuses on novel mechanisms involved in the crosstalk among epigenetic, signaling, transcriptional and metabolic pathways in TAMs. Particular attention has been given to the recently identified link between cancer cell metabolism and the epigenetic programming of TAMs by histone lactylation, which can be responsible for the unlimited protumoral programming of TAMs. Finally, we explain how TAMs interfere with currently used anticancer therapeutics and summarize the most advanced data from clinical trials, which we divide into four categories: inhibition of TAM survival and differentiation, inhibition of monocyte/TAM recruitment into tumors, functional reprogramming of TAMs, and genetic enhancement of macrophages.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


