STK19 通过刺激修复复合物的稳定性、RNA Pol II 泛素化和 TFIIH 招募来驱动转录耦合修复
IF 14.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
转录耦合核苷酸切除修复(TC-NER)可有效消除阻碍 RNA 聚合酶 II(RNA Pol II)进行基因转录的 DNA 损伤。TC-NER 由 CSB 识别损伤停滞的 RNA Pol II 启动,CSB 会招募 CRL4CSA 泛素连接酶和 UVSSA。CRL4CSA 在 RPB1-K1268 处的 RNA Pol II 泛素化是一个关键的 TC-NER 检查点,可控制 RNA Pol II 的稳定性,并通过 TFIIH 招募启动 DNA 损伤切除。然而,CRL4CSA 活性和 TFIIH 招募的精确调控机制仍然难以捉摸。在这里,我们揭示了人类丝氨酸/苏氨酸蛋白激酶19(STK19)作为TC-NER因子,对正确的DNA损伤切除和随后的转录重启至关重要。低温电子显微镜(cryo-EM)研究表明,STK19是RNA Pol II-TC-NER复合物的一个组成部分,它连接着CSA、UVSSA、RNA Pol II和下游DNA。STK19 能刺激 TC-NER 复合物的稳定性和 CRL4CSA 的活性,从而高效地实现 RNA Pol II 泛素化和正确的 UVSSA 与 TFIIH 结合。这些发现强调了 STK19 作为 TC-NER 核心部件的关键作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
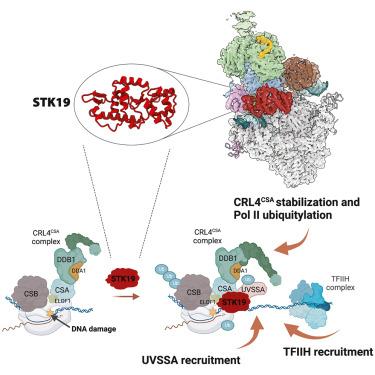
STK19 drives transcription-coupled repair by stimulating repair complex stability, RNA Pol II ubiquitylation, and TFIIH recruitment
Transcription-coupled nucleotide excision repair (TC-NER) efficiently eliminates DNA damage that impedes gene transcription by RNA polymerase II (RNA Pol II). TC-NER is initiated by the recognition of lesion-stalled RNA Pol II by CSB, which recruits the CRL4CSA ubiquitin ligase and UVSSA. RNA Pol II ubiquitylation at RPB1-K1268 by CRL4CSA serves as a critical TC-NER checkpoint, governing RNA Pol II stability and initiating DNA damage excision by TFIIH recruitment. However, the precise regulatory mechanisms of CRL4CSA activity and TFIIH recruitment remain elusive. Here, we reveal human serine/threonine-protein kinase 19 (STK19) as a TC-NER factor, which is essential for correct DNA damage removal and subsequent transcription restart. Cryogenic electron microscopy (cryo-EM) studies demonstrate that STK19 is an integral part of the RNA Pol II-TC-NER complex, bridging CSA, UVSSA, RNA Pol II, and downstream DNA. STK19 stimulates TC-NER complex stability and CRL4CSA activity, resulting in efficient RNA Pol II ubiquitylation and correct UVSSA and TFIIH binding. These findings underscore the crucial role of STK19 as a core TC-NER component.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Cell
生物-生化与分子生物学
CiteScore
26.00
自引率
3.80%
发文量
389
审稿时长
1 months
期刊介绍:
Molecular Cell is a companion to Cell, the leading journal of biology and the highest-impact journal in the world. Launched in December 1997 and published monthly. Molecular Cell is dedicated to publishing cutting-edge research in molecular biology, focusing on fundamental cellular processes. The journal encompasses a wide range of topics, including DNA replication, recombination, and repair; Chromatin biology and genome organization; Transcription; RNA processing and decay; Non-coding RNA function; Translation; Protein folding, modification, and quality control; Signal transduction pathways; Cell cycle and checkpoints; Cell death; Autophagy; Metabolism.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


