氮空位诱导的 n-π* 电子转变增强了氮化碳的光催化制氢能力
IF 4.1
3区 医学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
在半导体催化剂中,长寿命激发态可有效提高光生载流子的利用率,从而增强光催化性能。在此,我们利用超分子工程合成了一种具有 N 空位和明显 n-π* 转变的空心管状氮化碳催化剂。独特的空心管状结构提供了丰富的活性位点,有利于光催化反应。N 空位的存在扩大了共轭体系中的π电子析出域,激发了 n-π* 转变,从而引发了约 660 纳米波长处的红移吸收边。实验和 DFT 计算表明,N 空位有利于缩小带隙,促进光生电子还原 H+。飞秒瞬态吸收光谱(fs-TAS)表明,氮化碳光催化剂中的 n-π* 电子转变会导致更慢的激子湮灭(寿命:38.64 ± 10.6 ps)和更长的浅电子捕获态(寿命:325.9 ± 19.3 ps)。这些态的出现为光催化反应过程增加了更多的光生电子。最佳空心管状氮化碳催化剂的制氢率为 2664.47 μmol∙g-1∙h-1,是块状氮化碳催化剂(85.3325 μmol∙g-1∙h-1)的 31.2 倍。这项工作凸显了氮空位诱导的 n-π* 转变能够提高氮化碳的光催化活性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
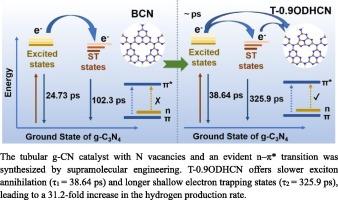
The n–π* electronic transition induced by nitrogen vacancies enhances photocatalytic hydrogen production in carbon nitride
In semiconductor catalysts, long-lived excited states can effectually improve the utilization of photogenerated carriers to enhance photocatalytic performance. Herein, we used supramolecular engineering to synthesize a hollow tubular carbon nitride catalyst with N vacancies and an obvious n–π* transition. The unique hollow tubular structure provides abundant active sites, which are favorable for photocatalytic reaction. The presence of N vacancies expands the π-electron delocalization domains in the conjugated system, which excites the n–π* transition and thus triggers the red-shifted absorption edge at approximately 660 nm. Experiments and DFT calculations demonstrated that the N vacancies are beneficial for narrowing the bandgap and promoting the reduction of H+ by photogenerated electrons. Femtosecond transient absorption spectroscopy (fs-TAS) indicated that the n–π* electronic transition in the carbon nitride photocatalyst leads to slower exciton annihilation (lifetime: 38.64 ± 10.6 ps) and extended shallow electron trapping states (lifetime: 325.9 ± 19.3 ps). The appearance of these states adds more photogenerated electrons to the photocatalytic reaction process. The optimal hollow tubular carbon nitride catalyst exhibits a hydrogen production rate of 2664.47 μmol∙g−1∙h−1, which is 31.2 times higher than that of bulk carbon nitride (85.3325 μmol∙g−1∙h−1). This work highlights the ability of the n–π* transition induced by N vacancies to enhance the photocatalytic activity of carbon nitride.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Chemical Neuroscience
BIOCHEMISTRY & MOLECULAR BIOLOGY-CHEMISTRY, MEDICINAL
CiteScore
9.20
自引率
4.00%
发文量
323
审稿时长
1 months
期刊介绍:
ACS Chemical Neuroscience publishes high-quality research articles and reviews that showcase chemical, quantitative biological, biophysical and bioengineering approaches to the understanding of the nervous system and to the development of new treatments for neurological disorders. Research in the journal focuses on aspects of chemical neurobiology and bio-neurochemistry such as the following:
Neurotransmitters and receptors
Neuropharmaceuticals and therapeutics
Neural development—Plasticity, and degeneration
Chemical, physical, and computational methods in neuroscience
Neuronal diseases—basis, detection, and treatment
Mechanism of aging, learning, memory and behavior
Pain and sensory processing
Neurotoxins
Neuroscience-inspired bioengineering
Development of methods in chemical neurobiology
Neuroimaging agents and technologies
Animal models for central nervous system diseases
Behavioral research
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


