PHLDA1 在炎症性肠病中通过限制肠上皮细胞凋亡保护肠屏障功能。
IF 3.3
3区 生物学
Q3 CELL BIOLOGY
引用次数: 0
摘要
目前治疗炎症性肠病(IBD)的方法主要侧重于控制炎症,而不是维持肠屏障的完整性。在我们的研究中,我们试图调查 PHLDA1 在维护肠屏障功能方面的潜在作用,以此作为治疗 IBD 的一种有前途的策略。我们观察到,在 IBD 患者和化学诱导结肠炎小鼠的肠上皮细胞(IECs)中,PHLDA1 的表达明显减少。PHLDA1 的缺乏导致 IEC 细胞凋亡增加,上皮屏障受损,共生细菌侵入粘膜。因此,与对照组小鼠相比,在 IECs 中特异性敲除 PHLDA1 的小鼠(Phlda1IEC-KO)体内微生物的入侵大大加剧了结肠炎症。从机理上讲,我们发现了 PHLDA1 与 MCL1 相互作用的证据,以保护 K48 链接的 K40 赖氨酸残基处的多泛素化,从而防止泛素蛋白酶体通过 MCL1 泛素连接酶 E3(Mule)降解。我们进一步证实,PHLDA1-MCL1-Mule 信号通路在 IBD 的发展中起着关键作用。值得注意的是,我们的研究表明,使用腺相关病毒(AAV)提高 MCL1 的水平或减少 Mule 的表达可减轻 Phlda1IEC-KO 小鼠的实验性结肠炎。总之,我们的研究结果强调了 PHLDA1 在 IBD 发病机制中的重要性,并提出靶向 PHLDA1-MCL1-Mule 信号通路可能是防治 IBD 的一种可行方法。本文章由计算机程序翻译,如有差异,请以英文原文为准。
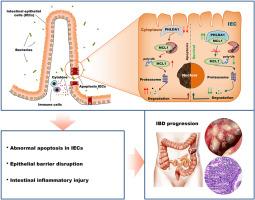
PHLDA1 protects intestinal barrier function via restricting intestinal epithelial cells apoptosis in inflammatory bowel disease
The current approach to treating inflammatory bowel disease (IBD) primarily focuses on managing inflammation rather than maintaining the integrity of the intestinal barrier. In our study, we sought to investigate the potential role of PHLDA1 in preserving intestinal barrier function as a promising strategy for treating IBD. We observed a significant decrease in PHLDA1 expression in intestinal epithelial cells (IECs) of both IBD patients and mice with chemically induced colitis. This deficiency of PHLDA1 led to increased apoptosis of IECs, resulting in a compromised epithelial barrier and the invasion of commensal bacteria into the mucosa. Consequently, this microbial invasion substantially exacerbated colonic inflammation in mice with the specific knockout of PHLDA1 in IECs (Phlda1IEC−KO) compared to their control littermates. Mechanistically, we found evidence of PHLDA1 interacting with MCL1 to protect against K48-linked polyubiquitylation at the K40 lysine residue, thus preventing ubiquitin-proteasome degradation through the MCL1 ubiquitin ligase E3 (Mule). We further confirmed that the PHLDA1-MCL1-Mule signaling pathway plays a critical role in the development of IBD. Notably, our study demonstrated that enhancing MCL1 levels or reducing Mule expression using adeno-associated virus (AAV) attenuated experimental colitis in Phlda1IEC−KO mice. Collectively, our findings emphasize the significance of PHLDA1 in the pathogenesis of IBD and propose that targeting the PHLDA1-MCL1-Mule signaling pathway could be a viable approach for combating IBD.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Experimental cell research
医学-细胞生物学
CiteScore
7.20
自引率
0.00%
发文量
295
审稿时长
30 days
期刊介绍:
Our scope includes but is not limited to areas such as: Chromosome biology; Chromatin and epigenetics; DNA repair; Gene regulation; Nuclear import-export; RNA processing; Non-coding RNAs; Organelle biology; The cytoskeleton; Intracellular trafficking; Cell-cell and cell-matrix interactions; Cell motility and migration; Cell proliferation; Cellular differentiation; Signal transduction; Programmed cell death.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


