观察作为苯的羰基硼类似物的芳香族 B13(CO)n+(n = 1-7
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
一氧化碳作为典型的σ-供体是化学中最重要的配体之一,而平面 B13+ 则是实验中最著名的类似于苯的魔数硼簇。本文进行的气相质谱、碰撞诱导解离和第一原理理论联合研究表明,在环境条件下,B13+ 与 CO 相继发生反应,形成一系列硼羰基复合物 B13(CO)n+,最高可达 n = 7,这是迄今为止观察到的最大的硼羰基复合物,其中心为准平面 B13+ 核心,周围由 nCO 配体配位。广泛的理论分析揭示了这些芳香族 B13(CO)n+ 单质的化学吸附途径和成键模式,这些单质在略微起皱的 B13+ 分子上保留了三个分散的 π 键,证明它们都是苯的硼羰基类似物,暂命名为硼羰基芳香族化合物 (BCA)。据预测,它们具有准平面 B12 配位中心的 B12(CO)n (n = 1-6)π-异价配合物将成为稳定的中性 BCAs。本文章由计算机程序翻译,如有差异,请以英文原文为准。
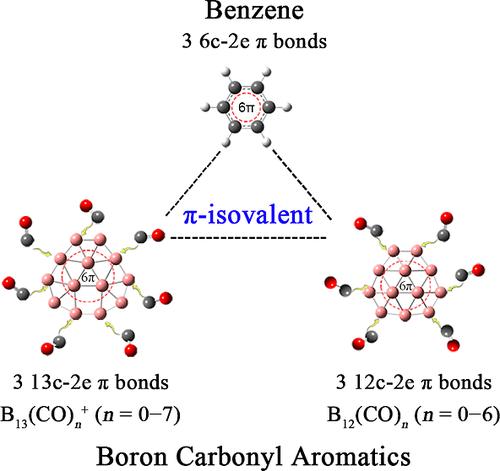
Observation of Aromatic B13(CO)n+ (n = 1–7) as Boron Carbonyl Analogs of Benzene
CO as a typical σ-donor is one of the most important ligands in chemistry, while planar B13+ is experimentally known as the most prominent magic-number boron cluster analogous to benzene. Joint gas-phase mass spectroscopy, collision-induced dissociation, and first-principles theory investigations performed herein indicate that B13+ reacts with CO successively under ambient conditions to form a series of boron carbonyl complexes B13(CO)n+ up to n = 7, presenting the largest boron carbonyl complexes observed to date with a quasi-planar B13+ core at the center coordinated by nCO ligands around it. Extensive theoretical analyses unveil both the chemisorption pathways and bonding patterns of these aromatic B13(CO)n+ monocations which, with three delocalized π bonds well-retained over the slightly wrinkled B13+ moiety, all prove to be boron carbonyl analogs of benzene tentatively named as boron carbonyl aromatics (BCAs). Their π-isovalent B12(CO)n (n = 1–6) complexes with a quasi-planar B12 coordination center are predicted to be stable neutral BCAs.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


