人脂肪细胞中 cAMP 驱动的 UCP1 诱导需要 ATGL 催化脂肪分解。
IF 7
2区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
摘要
目的:解偶联蛋白 1(UCP1)通过儿茶酚胺诱导的 cAMP 信号在棕色或 "米色 "脂肪细胞中被诱导,从而激活多种转录因子。PPARγ 激动剂如罗格列酮(Rsg)也能增强 UCP1 的表达。然而,目前还不清楚这种上调是源于米色脂肪细胞从祖细胞中重新分化,还是源于已存在的脂肪细胞对 UCP1 的诱导。为了探讨这个问题,我们使用了从祖细胞分化而来的人类脂肪细胞,并研究了它们对 Rsg、腺苷酸环化酶激活剂福斯可林(Fsk)或同时对这两种物质的急性反应:方法:在不接触 PPARγ 激动剂的情况下,对原代人类祖细胞产生的脂肪细胞进行分化,并对 Fsk、Rsg 或两者同时处理 3、6 或 78 小时。采用大量 RNASeq、RNAScope、RT-PCR、CRISPR-Cas9 介导的基因敲除、耗氧量和 Western 印迹法评估细胞反应:结果:UCP1 mRNA的表达在暴露于Rsg或Fsk后3小时内被诱导,这表明Rsg的作用与额外的脂肪细胞分化无关。尽管Rsg和Fsk诱导的整体转录反应不同,但两者都诱导了与钙代谢、脂滴组装和线粒体重塑相关的基因,这表明了人类脂肪细胞分化的核心特征。意想不到的是,我们发现在 CRISPR-Cas9 介导的 PNPLA2(编码甘油三酯脂肪酶 ATGL 的基因)敲除后,Fsk 诱导的 UCP1 表达减少了约 80%。正如预期的那样,ATGL基因敲除抑制了脂肪分解;然而,UCP1诱导的相关抑制表明,cAMP介导的最大UCP1诱导需要ATGL催化脂肪分解的产物。与此相佐证的是,我们观察到 ATGL 基因敲除导致的 Fsk 刺激 UCP1 诱导的减少被 Rsg 逆转,这意味着脂肪分解在此过程中的作用是产生天然 PPARγ 激动剂:结论:众所周知,UCP1 的转录是由 cAMP 依赖性蛋白激酶下游激活的转录因子刺激的。我们在此证明,UCP1 的转录也可通过 PPARγ 激活而被急性诱导。此外,在人类脂肪细胞中,这两种途径都会在 cAMP 作用下被激活,从而协同诱导 UCP1 的表达。cAMP 对 PPARγ 的刺激可能是通过 ATGL 介导的脂肪分解产生天然 PPARγ 激活配体的结果。本文章由计算机程序翻译,如有差异,请以英文原文为准。
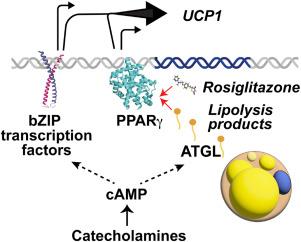
cAMP driven UCP1 induction in human adipocytes requires ATGL-catalyzed lipolysis
Objective
The uncoupling protein 1 (UCP1) is induced in brown or “beige” adipocytes through catecholamine-induced cAMP signaling, which activates diverse transcription factors. UCP1 expression can also be enhanced by PPARγ agonists such as rosiglitazone (Rsg). However, it is unclear whether this upregulation results from de-novo differentiation of beige adipocytes from progenitor cells, or from the induction of UCP1 in pre-existing adipocytes. To explore this, we employed human adipocytes differentiated from progenitor cells and examined their acute response to Rsg, to the adenylate-cyclase activator forskolin (Fsk), or to both simultaneously.
Methods
Adipocytes generated from primary human progenitor cells were differentiated without exposure to PPARγ agonists, and treated for 3, 6 or 78 h to Fsk, to Rsg, or to both simultaneously. Bulk RNASeq, RNAScope, RT-PCR, CRISPR-Cas9 mediated knockout, oxygen consumption and western blotting were used to assess cellular responses.
Results
UCP1 mRNA expression was induced within 3 h of exposure to either Rsg or Fsk, indicating that Rsg’s effect is independent on additional adipocyte differentiation. Although Rsg and Fsk induced distinct overall transcriptional responses, both induced genes associated with calcium metabolism, lipid droplet assembly, and mitochondrial remodeling, denoting core features of human adipocyte beiging. Unexpectedly, we found that Fsk-induced UCP1 expression was reduced by approximately 80% following CRISPR-Cas9-mediated knockout of PNPLA2, the gene encoding the triglyceride lipase ATGL. As anticipated, ATGL knockout suppressed lipolysis; however, the associated suppression of UCP1 induction indicates that maximal cAMP-mediated UCP1 induction requires products of ATGL-catalyzed lipolysis. Supporting this, we observed that the reduction in Fsk-stimulated UCP1 induction caused by ATGL knockout was reversed by Rsg, implying that the role of lipolysis in this process is to generate natural PPARγ agonists.
Conclusions
UCP1 transcription is known to be stimulated by transcription factors activated downstream of cAMP-dependent protein kinases. Here we demonstrate that UCP1 transcription can also be acutely induced through PPARγ-activation. Moreover, both pathways are activated in human adipocytes in response to cAMP, synergistically inducing UCP1 expression. The stimulation of PPARγ in response to cAMP may result from the production of natural PPARγ activating ligands through ATGL-mediated lipolysis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Metabolism
ENDOCRINOLOGY & METABOLISM-
CiteScore
14.50
自引率
2.50%
发文量
219
审稿时长
43 days
期刊介绍:
Molecular Metabolism is a leading journal dedicated to sharing groundbreaking discoveries in the field of energy homeostasis and the underlying factors of metabolic disorders. These disorders include obesity, diabetes, cardiovascular disease, and cancer. Our journal focuses on publishing research driven by hypotheses and conducted to the highest standards, aiming to provide a mechanistic understanding of energy homeostasis-related behavior, physiology, and dysfunction.
We promote interdisciplinary science, covering a broad range of approaches from molecules to humans throughout the lifespan. Our goal is to contribute to transformative research in metabolism, which has the potential to revolutionize the field. By enabling progress in the prognosis, prevention, and ultimately the cure of metabolic disorders and their long-term complications, our journal seeks to better the future of health and well-being.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


