水蒸发蛋白参与滋贺毒素诱导的人肾微血管内皮细胞肿胀和水转运功能障碍。
IF 4.6
2区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
Biochimica et biophysica acta. Molecular cell research
Pub Date : 2024-10-21
DOI:10.1016/j.bbamcr.2024.119866
引用次数: 0
摘要
产志贺毒素大肠埃希菌相关溶血性尿毒症(STEC-HUS)的特征之一是肾脏损伤。我们之前的研究表明,2 型志贺毒素(Stx2a)会降低细胞活力,并诱导人肾小球内皮细胞(HGEC)肿胀。然而,Stx2a 能破坏 HGEC 单层细胞的净水运输,而不影响细胞活力。这项研究旨在阐明 Stx2a 在 HGEC 单层细胞上造成水转运中断的可能机制。我们通过分析 FITC-二聚体的通过量、静水压(Pydr)和渗透压(Posm),分别研究了 HGEC 的胞旁和跨细胞水转运。Stx2a 选择性地影响跨细胞途径,而不影响旁细胞途径。此外,在使用水蒸发蛋白抑制剂四乙基氯化铵(TEA)、氯化汞(HgCl2)或 TGN-020 进行预处理后,Stx2a 细胞肿胀也会被阻止,这表明水蒸发蛋白参与了这一过程。共聚焦显微镜显示,Stx2a 增加了 HGEC 的总体积,而 TEA 和 TGN-020 则抵消了这一作用。此外,我们在 HGEC 中不仅发现了水通道蛋白-1(AQP1)的表达,还发现了水通道蛋白-4(AQP4)的表达。令人惊讶的是,在接触 Stx2a 后,我们观察到这两种 AQPs 的表达都有所下降。我们的研究结果表明,Stx2a 可能会诱导水分通过 AQP1 和 AQP4 进入 HGEC,从而增加细胞的总体积。随后,AQP1 和 AQP4 表达的减少可能会抑制跨细胞的水分转移,这可能是防止水分过度进入和细胞溶解的一种保护机制。本文章由计算机程序翻译,如有差异,请以英文原文为准。
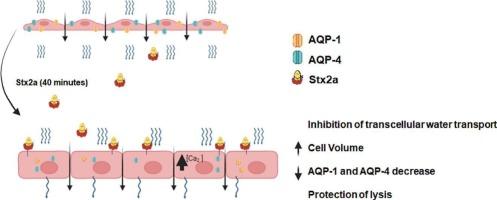
Involvement of aquaporins in Shiga toxin-induced swelling and water transport dysfunction in human renal microvascular endothelial cells
One of the hallmarks of Shiga toxin-producing Escherichia coli-associated hemolytic uremic syndrome (STEC-HUS) is kidney damage. Our previous research demonstrated that Shiga toxin type 2 (Stx2a) decreases cell viability and induces swelling of human glomerular endothelial cells (HGEC). However, Stx2a can disrupt net water transport across HGEC monolayers without affecting cell viability. This work aimed to elucidate the possible mechanisms involved in the water transport disruption caused by Stx2a across HGEC monolayers. We investigated paracellular and transcellular water transfer across HGEC by analyzing the passage of FITC-Dextran and the hydrostatic pressure (Phydr) and measuring the osmotic pressure (Posm), respectively. Stx2a selectively affected the transcellular pathway without impacting the paracellular route. Furthermore, Stx2a cell swelling was prevented by pretreatment with aquaporin inhibitors tetraethylammonium chloride (TEA), Mercury (II) chloride (HgCl2) or TGN-020, suggesting aquaporin involvement in this process. Confocal microscopy revealed that Stx2a increased HGEC total volume, which TEA and TGN-020 counteracted. Additionally, we identified in HGEC not only the expression of aquaporin-1 (AQP1) but also the expression of aquaporin-4 (AQP4). Surprisingly, we observed a decrease in the expression of both AQPs after Stx2a exposure. Our findings suggest that Stx2a may induce water movement into HGEC via AQP1 and AQP4, increasing total cell volume. Subsequently, decreased AQP1 and AQP4 expression could inhibit transcellular water transfer, potentially as a protective mechanism against excessive water entry and cell lysis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
10.00
自引率
2.00%
发文量
151
审稿时长
44 days
期刊介绍:
BBA Molecular Cell Research focuses on understanding the mechanisms of cellular processes at the molecular level. These include aspects of cellular signaling, signal transduction, cell cycle, apoptosis, intracellular trafficking, secretory and endocytic pathways, biogenesis of cell organelles, cytoskeletal structures, cellular interactions, cell/tissue differentiation and cellular enzymology. Also included are studies at the interface between Cell Biology and Biophysics which apply for example novel imaging methods for characterizing cellular processes.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


