α-环糊精水合物中水的不同状态
IF 2.2
3区 工程技术
Q3 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
α-CD-nH2O 水合物上的饱和和不饱和水蒸气压力是在完全失水的条件下用膜规压力计通过静态方法测量的。获得的数据表明,α-CD-nH2O 水合物中存在挥发性不同的水分子。挥发性较低的水分子可被视为 "外部 "水分子,包含在分子间氢键网络中,而挥发性较高的水分子(在我们之前的研究中发现)则被视为 "内部 "水分子,可能位于 α-CD 的空腔中。对于每种成分,我们都确定了 "外部 "和 "内部 "水的含量,并得到了描述α-CD-nH2O 中各类水的含量与 n 的关系的方程。计算了 lnp - 1/T 相关性的分析表达式以及所研究过程的焓值和熵值。实验数据用于获得 "外部 "水分子与 α-CD 框架结合时的吉布斯能变化。所得数值与我们之前研究中给出的 "内部 "水的数值有所不同。本研究还观察到了之前研究中发现的 α-CD-nH2O 中的相变,其热力学特征与之前的研究结果非常吻合。1H NMR 研究也证实了这些发现。本文章由计算机程序翻译,如有差异,请以英文原文为准。
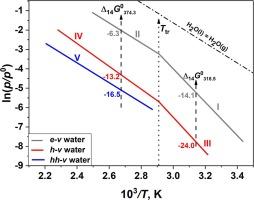
Different States of water in α-Cyclodextrin hydrates
The pressure of saturated and unsaturated water vapor over α-CD·nH2O hydrates has been measured by static method with membrane-gauge manometer under conditions of complete loss of water. The temperature dependences of water vapor pressure have been obtained in the wide range of temperatures (287–469 K), pressures (0.04–34.95 kPa) and water content (0.17 ≤ n ≤ 6.0).
The data obtained indicate the presence of water molecules with different volatility in α-CD·nH2O hydrates. Water molecules with lower volatility can be considered “external”, included in the network of intermolecular hydrogen bonds, while water molecules with higher volatility, discovered in our previous study, are considered “internal”, presumably located in the α-CD cavity. For each composition, the amount of “external” and “internal” water has been determined and the equations described the dependence of the content of each type of water on n in α-CD·nH2O have been obtained. The analytical expressions for the lnp – 1/T dependences as well as the values of enthalpy and entropy of processes studied have been calculated. Experimental data were used to obtain the Gibbs energy changes when “external” water molecules bind to the α-CD framework. The values obtained are differed from the values for “internal” water given in our previous study. The phase transition in α-CD·nH2O revealed in our previous study is also observed in this work and its thermodynamic characteristics are in good agreement with the results obtained earlier. The findings have been confirmed by 1H NMR studies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Chemical Thermodynamics
工程技术-热力学
CiteScore
5.60
自引率
15.40%
发文量
199
审稿时长
79 days
期刊介绍:
The Journal of Chemical Thermodynamics exists primarily for dissemination of significant new knowledge in experimental equilibrium thermodynamics and transport properties of chemical systems. The defining attributes of The Journal are the quality and relevance of the papers published.
The Journal publishes work relating to gases, liquids, solids, polymers, mixtures, solutions and interfaces. Studies on systems with variability, such as biological or bio-based materials, gas hydrates, among others, will also be considered provided these are well characterized and reproducible where possible. Experimental methods should be described in sufficient detail to allow critical assessment of the accuracy claimed.
Authors are encouraged to provide physical or chemical interpretations of the results. Articles can contain modelling sections providing representations of data or molecular insights into the properties or transformations studied. Theoretical papers on chemical thermodynamics using molecular theory or modelling are also considered.
The Journal welcomes review articles in the field of chemical thermodynamics but prospective authors should first consult one of the Editors concerning the suitability of the proposed review.
Contributions of a routine nature or reporting on uncharacterised materials are not accepted.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


