SCF3 烯烃的立体异构氢卤化和交叉耦合反应
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
SCF3 取代的内炔经过区域和立体选择性氢卤化反应,生成相应的 β-卤代-α-SCF3-烯。SCF3-alkynes 中 C≡C 三键的强极化确保了区域选择性,而立体选择性则受质子供体的强度和/或溶剂极性的影响。β-碘-α-SCF3-烯烃的潜力在几个交叉耦合、原脱碘和氰化反应中得到了说明。本文章由计算机程序翻译,如有差异,请以英文原文为准。
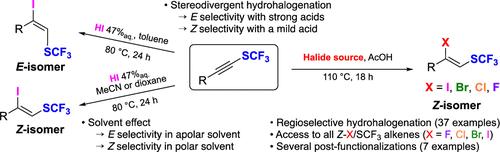
Stereodivergent Hydrohalogenation of SCF3 Alkynes and Cross- Coupling Reactions
SCF3-substituted internal alkynes undergo regio- and stereoselective hydrohalogenation to provide the corresponding β-halo-α-SCF3-alkenes. The regioselectivity is ensured by the strong polarization of the C≡C triple bond in SCF3-alkynes, while the stereoselectivity is governed by the strength of the proton donor and/or by the solvent polarity. The potential of β-iodo-α-SCF3-alkenes was illustrated in several cross-coupling, protodeiodination, and cyanation reactions.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


