金鸡纳有机催化的对映体选择性胺化季铵盐化丝氨酸作为叔酰胺
IF 5
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
在此,我们介绍了一种由 Cinchona-aminocatalyzed 对映体选择性 α-hydrazination 催化的 α-甲酰基酰胺化反应,用于生产作为叔酰胺的受保护季铵化丝氨酸,ee's 一般为 98%,收率≤99%。密度泛函理论计算支持的拟议 TS 模型涉及喹啉离子布氏酸辅助 DtBAD 的传递,当使用 9S- 腙胺催化剂时,DtBAD 从 H 键烯丙酮的 Re 面发生传递,从而产生一种通过 X 射线分析确定为 R 构型的酰肼。本文章由计算机程序翻译,如有差异,请以英文原文为准。
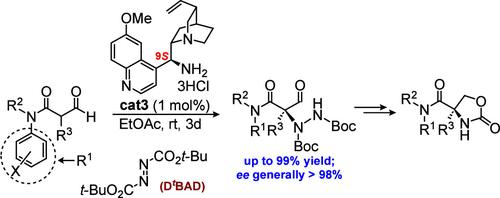
Cinchona Organocatalyzed Enantioselective Amination for Quaternized Serines as Tertiary Amides
Herein, we describe a Cinchona-aminocatalyzed enantioselective α-hydrazination of an α-formyl amide for the production of protected quaternized serines as tertiary amides with ee’s of generally >98% and ≤99% yields. The proposed TS model supported by density functional theory calculations involves a quinuclidinium ion Brønsted acid-assisted delivery of DtBAD, which occurs from the Re face of an H-bonded enaminone when using a 9S-cinchonamine catalyst, resulting in a hydrazide with the R-configuration as determined by X-ray analysis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


