通过非活化烯的电化学还原加氢反应实现二元环化/螺环化
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
本文介绍了一种通过还原脱芳烃化对一系列非活化芳烃(包括 N-取代吲哚、吲哚-3-甲酰胺衍生物和碘代苯甲酰胺)进行电化学环化/螺环化加氢反应的方法。该方法具有原子效率高、底物适用性广和选择性好等优点。利用一个简单的未分裂单元,通过芳基与芳香环的分子内加成,各种非活化烷烃发生环化/螺环化反应,生成 50 种吲哚类化合物、螺环化加氢产物和菲啶酮类化合物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
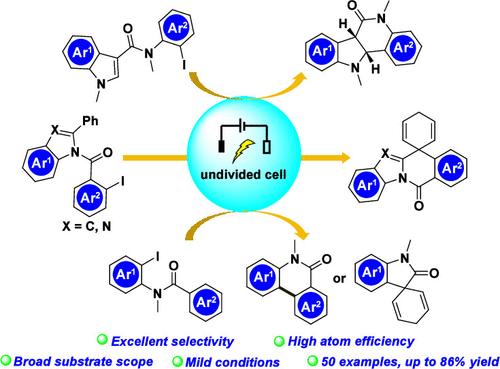
Dearomative Cyclization/Spirocyclization via Electrochemical Reductive Hydroarylation of Nonactivated Arenes
An electrochemical cyclization/spirocyclization hydroarylation via reductive dearomatization of a series of nonactivated arenes including N-substituted indoles, indole-3-carboxamide derivatives, and iodo-substituted benzamides is described. This protocol boasts high atom efficiency, broad substrate applicability, and excellent selectivity. Utilizing a simple undivided cell, various nonactivated arenes undergo cyclization/spirocyclization through the intramolecular addition of aryl radicals to an aromatic ring, yielding 50 indolines, spirocyclizative hydroarylation products, and phenanthridinones.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


