帕拉卡西酵母细胞外囊泡可改善高氨血症大鼠的神经炎症、GABA 神经传导和运动不协调。
IF 8.8
2区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
肝硬化患者可能会出现轻度肝性脑病(MHE),伴有运动不协调和认知障碍,从而降低生活质量和寿命。运动不协调是由于神经炎症和小脑GABA能神经传递增强所致。最近的报告证实,益生菌(包括干酪乳杆菌)可改善不同病理情况下的认知功能和肝硬化患者的 MHE。细胞外囊泡(EV)是源自细胞的小膜泡,携带从细胞(包括细菌)释放的生物活性分子。我们假设,来自副酸性乳杆菌(LC-EV)的EV可以改善MHE的神经炎症、GABA能神经传递和运动功能。我们的研究表明,LC-EV 治疗可逆转高氨血症大鼠小脑胶质细胞的激活和神经炎症,并恢复其运动协调能力。此外,用 LC-EV 对高氨血症大鼠的小脑切片进行体外治疗也能逆转神经胶质细胞的激活和神经炎症,并增强 TNFR1-S1PR2-BDNF-TrkB 和 TNFR1-TrkB-pAKT-NFκB -谷氨酰胺酶-GAT3 通路以及 GABA 能神经传递。报告的结果证明,LC-EV 可作为一种治疗工具,用于改善 MHE 患者的运动不协调。本文章由计算机程序翻译,如有差异,请以英文原文为准。
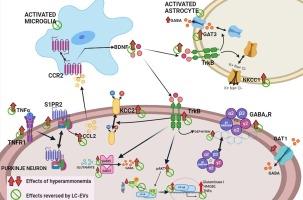
Extracellular vesicles from L. paracasei improve neuroinflammation, GABA neurotransmission and motor incoordination in hyperammonemic rats
Patients with liver cirrhosis may show minimal hepatic encephalopathy (MHE) with motor incoordination and cognitive impairment that reduce life quality and span. Motor incoordination is due to neuroinflammation and enhanced GABAergic neurotransmission in cerebellum. Recent reports support that probiotics, including L. casei, may improve cognitive function in different pathologies and MHE in cirrhotic patients. Extracellular vesicles (EV) are small cell-derived membrane vesicles that carry bioactive molecules released from cells, including bacteria. We hypothesized that EV from Lacticaseibacillus paracasei (LC-EV) could improve neuroinflammation, GABAergic neurotransmission and motor function in MHE. We show that LC-EV treatment reverses glial activation and neuroinflammation in cerebellum and restore motor coordination in hyperammonemic rats. Moreover, ex vivo treatment of cerebellar slices from hyperammonemic rats with LC-EV also reverses glial activation and neuroinflammation, and the enhancement of the TNFR1-S1PR2-BDNF-TrkB and TNFR1-TrkB-pAKT-NFκB-glutaminase-GAT3 pathways and of GABAergic neurotransmission. The results reported support that LC-EV may be used as a therapeutic tool to improve motor incoordination in patients with MHE.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
29.60
自引率
2.00%
发文量
290
审稿时长
28 days
期刊介绍:
Established in 1987, Brain, Behavior, and Immunity proudly serves as the official journal of the Psychoneuroimmunology Research Society (PNIRS). This pioneering journal is dedicated to publishing peer-reviewed basic, experimental, and clinical studies that explore the intricate interactions among behavioral, neural, endocrine, and immune systems in both humans and animals.
As an international and interdisciplinary platform, Brain, Behavior, and Immunity focuses on original research spanning neuroscience, immunology, integrative physiology, behavioral biology, psychiatry, psychology, and clinical medicine. The journal is inclusive of research conducted at various levels, including molecular, cellular, social, and whole organism perspectives. With a commitment to efficiency, the journal facilitates online submission and review, ensuring timely publication of experimental results. Manuscripts typically undergo peer review and are returned to authors within 30 days of submission. It's worth noting that Brain, Behavior, and Immunity, published eight times a year, does not impose submission fees or page charges, fostering an open and accessible platform for scientific discourse.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


