纳米盘相关乙酰胆碱酯酶作为生理相关膜结合胆碱酯酶的新型模型系统。酚类化合物的抑制作用。
IF 2.5
3区 生物学
Q3 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
乙酰胆碱酯酶(AChE)在胆碱能系统中起着举足轻重的作用,从昆虫防治到阿尔茨海默病治疗,人们都在寻求抑制乙酰胆碱酯酶的方法。虽然 AChE 的主要生理异构体是膜结合蛋白,但大多数发现新的、更安全、更有效的抑制剂的试验都是使用市售的可溶性异构体(如电鳗 AChE(eeAChE))进行的。在这项研究中,我们对重组人 AChE、eeAChE 和一种称为 dAChE4 的人 AChE 突变变体的酚类抑制剂的活性和选择性进行了比较分析。尽管存在许多突变,但 dAChE4 与其亲本蛋白非常相似,是人类 AChE 单体的合适模型。我们还建立了一个膜结合 AChE 体外系统,以创建一个近似生理异构体的模型。该系统确保了酶的正常工作,并使我们能够精确控制每次测定的酶和脂质浓度。本文章由计算机程序翻译,如有差异,请以英文原文为准。
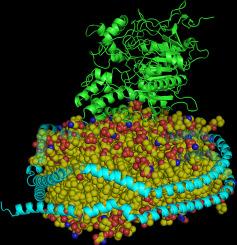
Nanodisc-associated acetylcholinesterase as a novel model system of physiological relevant membrane-bound cholinesterases. Inhibition by phenolic compounds
Acetylcholinesterase (AChE) plays a pivotal role in the cholinergic system, and its inhibition is sought after in a wide range of applications, from insect control to Alzheimer's disease treatment. While the primary physiological isoforms of AChE are membrane-bound proteins, most assays for discovering new, safer, and potent inhibitors are conducted using commercially available soluble isoforms, such as the electric eel AChE (eeAChE). In this study, we conducted a comparative analysis of the activity and selectivity to phenolic inhibitors of recombinant human AChE, eeAChE and a mutant variant of human AChE known as dAChE4. Despite numerous mutations, dAChE4 closely resembles its parental protein and serves as a suitable model for monomeric human AChE. We also established an in vitro system of membrane-bound AChE to create a model that closely mimics the physiological isoforms. This system ensures the proper work of the enzyme and allowed us to control the exact concentration of enzyme and lipids per assay.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Biochimica et biophysica acta. Biomembranes
生物-生化与分子生物学
CiteScore
8.20
自引率
5.90%
发文量
175
审稿时长
2.3 months
期刊介绍:
BBA Biomembranes has its main focus on membrane structure, function and biomolecular organization, membrane proteins, receptors, channels and anchors, fluidity and composition, model membranes and liposomes, membrane surface studies and ligand interactions, transport studies, and membrane dynamics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


