BRCA1水平和DNA损伤反应受控于circHIPK3或FMRP与BRCA1 mRNA的竞争性结合
IF 14.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
环状 RNA(circRNA)是在真核生物中广泛表达的共价闭合 RNA 分子,在包括癌症在内的多种病理过程中都会发生失调。许多研究指出它们具有作为微RNA(miRNA)和蛋白质海绵的活性;然而,我们提出了一种基于circRNA-mRNA相互作用的功能,以调控mRNA的命运。我们发现,在人类癌细胞中,与肿瘤广泛相关的 circHIPK3 通过反向剪接区与 BRCA1 mRNA 直接相互作用。这种相互作用通过竞争fragile-X mental retardation 1 protein (FMRP)蛋白的结合来增加BRCA1的翻译。CircHIPK3耗竭或circRNA-mRNA相互作用中断会降低BRCA1蛋白水平并增加DNA损伤,使一些癌细胞对DNA损伤诱导剂敏感,并使它们易受合成致死的影响。此外,用锁定核酸(LNA)阻断 FMRP 与 BRCA1 mRNA 的相互作用可恢复 BRCA1 半合子乳腺癌细胞的生理性蛋白水平,这突出了 circRNA-mRNA 相互作用在调节 DNA 损伤反应中的重要性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
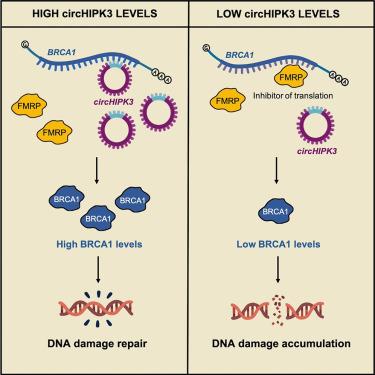
BRCA1 levels and DNA-damage response are controlled by the competitive binding of circHIPK3 or FMRP to the BRCA1 mRNA
Circular RNAs (circRNAs) are covalently closed RNA molecules widely expressed in eukaryotes and deregulated in several pathologies, including cancer. Many studies point to their activity as microRNAs (miRNAs) and protein sponges; however, we propose a function based on circRNA-mRNA interaction to regulate mRNA fate. We show that the widely tumor-associated circHIPK3 directly interacts in vivo with the BRCA1 mRNA through the back-splicing region in human cancer cells. This interaction increases BRCA1 translation by competing for the binding of the fragile-X mental retardation 1 protein (FMRP) protein, which we identified as a BRCA1 translational repressor. CircHIPK3 depletion or disruption of the circRNA-mRNA interaction decreases BRCA1 protein levels and increases DNA damage, sensitizing several cancer cells to DNA-damage-inducing agents and rendering them susceptible to synthetic lethality. Additionally, blocking FMRP interaction with BRCA1 mRNA with locked nucleic acid (LNA) restores physiological protein levels in BRCA1 hemizygous breast cancer cells, underscoring the importance of this circRNA-mRNA interaction in regulating DNA-damage response.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Cell
生物-生化与分子生物学
CiteScore
26.00
自引率
3.80%
发文量
389
审稿时长
1 months
期刊介绍:
Molecular Cell is a companion to Cell, the leading journal of biology and the highest-impact journal in the world. Launched in December 1997 and published monthly. Molecular Cell is dedicated to publishing cutting-edge research in molecular biology, focusing on fundamental cellular processes. The journal encompasses a wide range of topics, including DNA replication, recombination, and repair; Chromatin biology and genome organization; Transcription; RNA processing and decay; Non-coding RNA function; Translation; Protein folding, modification, and quality control; Signal transduction pathways; Cell cycle and checkpoints; Cell death; Autophagy; Metabolism.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


