HKDC1 作为葡萄糖传感器发挥作用,通过与 PHB2 相互作用促进新陈代谢适应和癌症生长
IF 15.4
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
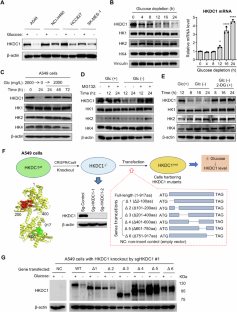
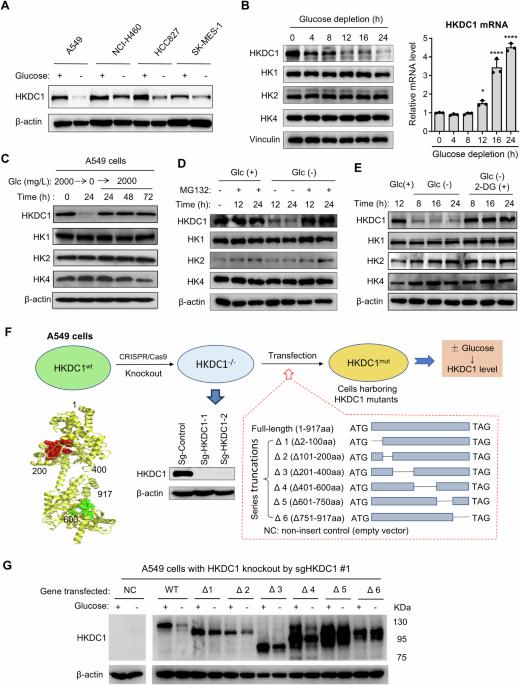
葡萄糖感应和代谢适应肿瘤微环境中的葡萄糖供应对癌症的发展至关重要。我们在这里发现,HKDC1 是一种在预后不良的癌症中高表达的六磷酸酶,它具有葡萄糖传感器的功能,会随着环境葡萄糖的变化而改变其稳定性。葡萄糖感应结构域位于 751-917 个氨基酸之间,其中 Ser896 是一个关键残基,它通过影响 Lys620 泛素化来调节 HKDC1 的稳定性。这种传感机制通过促进线粒体脂肪酸的利用,使细胞适应葡萄糖饥饿。此外,HKDC1 还通过封存禁止素 2 (PHB2),使其失去对 SP1 的抑制作用,从而促进促癌分子的表达,从而促进肿瘤生长。通过基因敲除或葡萄糖耗竭来废除 HKDC1 可释放 PHB2,从而抑制癌细胞增殖并抑制肿瘤生长。我们的研究揭示了 HKDC1 在葡萄糖感知和代谢适应中的作用,并将 HKDC1 确定为潜在的治疗靶点。本文章由计算机程序翻译,如有差异,请以英文原文为准。
HKDC1 functions as a glucose sensor and promotes metabolic adaptation and cancer growth via interaction with PHB2
Glucose sensing and metabolic adaptation to glucose availability in the tumor microenvironment are critical for cancer development. Here we show that HKDC1, a hexokinase highly expressed in cancer associated with poor prognosis, functions as a glucose sensor that alters its stability in response to environmental glucose. The glucose-sensing domain is located between amino acids 751-917, with Ser896 as a key residue that regulates HKDC1 stability by affecting Lys620 ubiquitination. This sensing mechanism enables cellular adaptation to glucose starvation by promoting mitochondrial fatty acid utilization. Furthermore, HKDC1 promotes tumor growth by sequestering prohibitin 2 (PHB2) to disable its suppressive effect on SP1, thus promoting the expression of pro-oncogenic molecules. Abrogation of HKDC1 by genetic knockout or by glucose depletion releases PHB2, leading to suppression of cancer cell proliferation and inhibition of tumor growth. Our study reveals a previously unrecognized role of HKDC1 in glucose sensing and metabolic adaptation, and identifies HKDC1 as a potential therapeutic target.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell Death and Differentiation
生物-生化与分子生物学
CiteScore
24.70
自引率
1.60%
发文量
181
审稿时长
3 months
期刊介绍:
Mission, vision and values of Cell Death & Differentiation:
To devote itself to scientific excellence in the field of cell biology, molecular biology, and biochemistry of cell death and disease.
To provide a unified forum for scientists and clinical researchers
It is committed to the rapid publication of high quality original papers relating to these subjects, together with topical, usually solicited, reviews, meeting reports, editorial correspondence and occasional commentaries on controversial and scientifically informative issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


