药物抗坏血酸、吉西他滨和纳布-紫杉醇治疗转移性胰腺癌的随机试验。
IF 10.7
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
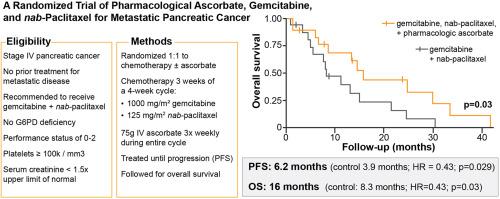
背景转移性胰腺导管腺癌(PDAC)患者的 5 年生存率很低。药理抗坏血酸(P-AscH-,大剂量、静脉注射、维生素 C)作为化疗的辅助药物已显示出前景。我们假设在吉西他滨和纳布紫杉醇中加入 P-AscH- 会提高转移性 PDAC 患者的生存率:被诊断为 IV 期胰腺癌的患者按 1:1 随机分配到只接受吉西他滨和纳布-紫杉醇治疗(SOC,对照组)或接受 SOC 并同时接受每周三次、每次 75 克的 P-AscH-治疗(ASC,研究组)。主要研究结果为总生存期,次要研究结果为无进展生存期和不良反应发生率。生活质量和患者对常见肿瘤症状的报告结果是一项探索性目标。36名参与者被随机分配,其中34人接受了指定的研究治疗。所有分析均基于2023年12月11日冻结的数据:静脉注射 P-AscH- 可使血清抗坏血酸水平从微摩尔级升至毫摩尔级。在吉西他滨+纳布-紫杉醇(ASC)中加入P-AscH-可将总生存期延长至16个月,而吉西他滨+纳布-紫杉醇(SOC)的总生存期仅为8.3个月(HR = 0.46; 90 % CI 0.23, 0.92; p = 0.030)。中位无进展生存期为6.2个月(ASC)对3.9个月(SOC)(HR = 0.43; 90 % CI 0.20, 0.92; p = 0.029)。添加 P-AscH- 不会对生活质量产生负面影响,也不会增加不良事件的频率或严重程度:结论:每周三次输注 75 克 P-AscH- 可延长转移性胰腺癌患者的总生存期和无进展生存期,且不会影响生活质量或增加毒性(ClinicalTrials.gov 编号 NCT02905578)。本文章由计算机程序翻译,如有差异,请以英文原文为准。
A randomized trial of pharmacological ascorbate, gemcitabine, and nab-paclitaxel for metastatic pancreatic cancer
Background
Patients with metastatic pancreatic ductal adenocarcinoma (PDAC) have poor 5-year survival. Pharmacological ascorbate (P-AscH-, high dose, intravenous, vitamin C) has shown promise as an adjunct to chemotherapy. We hypothesized adding P-AscH- to gemcitabine and nab-paclitaxel would increase survival in patients with metastatic PDAC.
Methods
Patients diagnosed with stage IV pancreatic cancer randomized 1:1 to gemcitabine and nab-paclitaxel only (SOC, control) or to SOC with concomitant P-AscH−, 75 g three times weekly (ASC, investigational). The primary outcome was overall survival with secondary objectives of determining progression-free survival and adverse event incidence. Quality of life and patient reported outcomes for common oncologic symptoms were captured as an exploratory objective. Thirty-six participants were randomized; of this 34 received their assigned study treatment. All analyses were based on data frozen on December 11, 2023.
Results
Intravenous P-AscH- increased serum ascorbate levels from micromolar to millimolar levels. P-AscH- added to the gemcitabine + nab-paclitaxel (ASC) increased overall survival to 16 months compared to 8.3 months with gemcitabine + nab-paclitaxel (SOC) (HR = 0.46; 90 % CI 0.23, 0.92; p = 0.030). Median progression free survival was 6.2 (ASC) vs. 3.9 months (SOC) (HR = 0.43; 90 % CI 0.20, 0.92; p = 0.029). Adding P-AscH- did not negatively impact quality of life or increase the frequency or severity of adverse events.
Conclusions
P-AscH− infusions of 75 g three times weekly in patients with metastatic pancreatic cancer prolongs overall and progression free survival without detriment to quality of life or added toxicity (ClinicalTrials.gov number NCT02905578).
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Redox Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-
CiteScore
19.90
自引率
3.50%
发文量
318
审稿时长
25 days
期刊介绍:
Redox Biology is the official journal of the Society for Redox Biology and Medicine and the Society for Free Radical Research-Europe. It is also affiliated with the International Society for Free Radical Research (SFRRI). This journal serves as a platform for publishing pioneering research, innovative methods, and comprehensive review articles in the field of redox biology, encompassing both health and disease.
Redox Biology welcomes various forms of contributions, including research articles (short or full communications), methods, mini-reviews, and commentaries. Through its diverse range of published content, Redox Biology aims to foster advancements and insights in the understanding of redox biology and its implications.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


