关于肌球蛋白方向性的控制
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
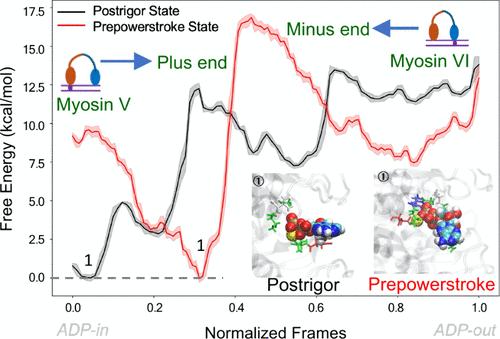
肌球蛋白独特方向性的起源一直是一个具有基础和实际意义的问题。这项工作以确凿的方式证明,肌球蛋白的方向性是通过调整决定速率步骤(即 ADP 释放步骤)的屏障来控制的。这一结论是在探索肌球蛋白 V 和 VI 反方向性背后的分子起源以及确定正向和反向运动 ADP 释放障碍变化的起源的基础上得出的。我们的研究结合了不同的模拟方法,如转向分子动力学(SMD)、伞状采样、重正化方法和自动路径搜索方法。研究发现,在肌球蛋白 V 的情况下,后冲程(后头部)状态下的 ADP 释放所克服的障碍比前冲程(前头部)状态下的低,这一点从实验观察中也可以看出。由于肌球蛋白 V 和 VI 的方向性趋势相反,我们得出结论,这种方向性差异是由 ADP 释放的自由能障碍控制的。总之,证明肌球蛋白的方向性是由循环中决定速率步骤的活化屏障决定的,而不是由某些未指定的动力学效应决定的,具有普遍意义。本文章由计算机程序翻译,如有差异,请以英文原文为准。
On the Control of Directionality of Myosin
The origin of the unique directionality of myosin has been a problem of fundamental and practical importance. This work establishes in a conclusive way that the directionality is controlled by tuning the barrier for the rate-determining step, namely, the ADP release step. This conclusion is based on exploring the molecular origin behind the reverse directionality of myosins V and VI and the determination of the origin of the change in the barriers of the ADP release for the forward and backward motions. Our investigation is performed by combining different simulation methods such as steer molecular dynamics (SMD), umbrella sampling, renormalization method, and automated path searching method. It is found that in the case of myosin V, the ADP release from the postrigor (trailing head) state overcomes a lower barrier than the prepowerstroke (leading head) state, which is also evident from experimental observation. In the case of myosin VI, we noticed a different trend when compared to myosin V. Since the directionality of myosins V and VI follows a reverse trend, we conclude that such differences in the directionality are controlled by the free energy barrier for the ADP release. Overall, the proof that the directionality of myosin is determined by the activation barrier of the rate-determining step in the cycle, rather than by some unspecified dynamical effects, has general importance.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


