TP53缺失的表型特征可预测对化疗的反应。
IF 6.8
1区 医学
Q1 ONCOLOGY
引用次数: 0
摘要
在临床前研究中,p53 功能缺失会影响化疗反应,但这一点尚未在临床上得到一致验证。我们从 TCGA 中的泛癌症临床样本中训练出了 TP53 缺失表型基因表达特征。在体外,TP53 缺失表型特征可预测不同癌症类型的化疗反应。在3003个接受新辅助化疗的乳腺癌样本的临床数据集中,TP53缺失表型样本获得病理完全反应(pCR)的可能性增加了56%,在ER阳性和ER阴性肿瘤中,TP53缺失表型与pCR之间都有显著的关联。在I-SPY2试验(N = 987)的独立临床验证中,我们证实了TP53缺失表型与新辅助化疗pCR的关系,并发现与非TP53缺失表型肿瘤相比,TP53缺失表型肿瘤的化疗免疫反应率更高(64%对28%)。TP53缺失表型特征在体外可预测各种癌症类型的化疗反应,在概念验证的临床验证中,它与多个临床乳腺癌队列的新辅助化疗反应相关。本文章由计算机程序翻译,如有差异,请以英文原文为准。
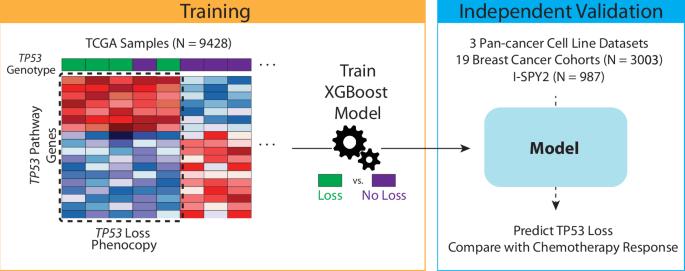
A phenocopy signature of TP53 loss predicts response to chemotherapy
In preclinical studies, p53 loss of function impacts chemotherapy response, but this has not been consistently validated clinically. We trained a TP53-loss phenocopy gene expression signature from pan-cancer clinical samples in the TCGA. In vitro, the TP53-loss phenocopy signature predicted chemotherapy response across cancer types. In a clinical dataset of 3003 breast cancer samples treated with neoadjuvant chemotherapy, the TP53-loss phenocopy samples were 56% more likely to have a pathologic complete response (pCR), with a significant association between TP53-loss phenocopy and pCR in both ER positive and ER negative tumors. In an independent clinical validation in the I-SPY2 trial (N = 987), we confirmed the association with neoadjuvant chemotherapy pCR and found higher rates of chemoimmunotherapy response in TP53-loss phenocopy tumors compared to non-TP53-loss phenocopy tumors (64% vs. 28%). The TP53-loss phenocopy signature predicts chemotherapy response across cancer types in vitro, and in a proof-of-concept clinical validation is associated with neoadjuvant chemotherapy response across multiple clinical breast cancer cohorts.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

NPJ Precision Oncology
ONCOLOGY-
CiteScore
9.90
自引率
1.30%
发文量
87
审稿时长
18 weeks
期刊介绍:
Online-only and open access, npj Precision Oncology is an international, peer-reviewed journal dedicated to showcasing cutting-edge scientific research in all facets of precision oncology, spanning from fundamental science to translational applications and clinical medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


