液相混合焓的数据驱动研究
IF 1.9
3区 材料科学
Q4 CHEMISTRY, PHYSICAL
Calphad-computer Coupling of Phase Diagrams and Thermochemistry
Pub Date : 2024-09-25
DOI:10.1016/j.calphad.2024.102745
引用次数: 0
摘要
液相混合焓是一种反映元素间相互作用的热力学性质,是预测相变的关键。目前已有广泛使用的模型来预测它,但从未对它们进行过系统评估。为了解决这个问题,我们从大约 1000 次热力学评估中收集了大量二元液体的混合焓数据。这使我们能够明确目前最先进的 Miedema 模型的预测精度。我们表明,基于 LightGBM 的机器学习模型可以获得更准确的预测结果,我们还提供了 2415 个二元体系的预测结果。我们收集的数据还允许我们对另一个预测过剩热容量的经验模型进行评估,该模型适用于 2211 种二元液体。然后,我们将数据收集扩展到三元金属液体,并发现当混合放热时,根据 Muggianu 模型从二元系统推断出的结果会在接近等摩尔组成时略微高估约 10%。因此,我们的 LightGBM 模型可以为三元合金提供合理的估计值,进而为多组分合金提供合理的估计值。我们从丰富的数据集中提取的研究结果可用于为材料开发的热力学、经验和机器学习模型提供支持。我们的数据、预测以及从热力学特性生成机器学习描述符的代码均已公布。本文章由计算机程序翻译,如有差异,请以英文原文为准。
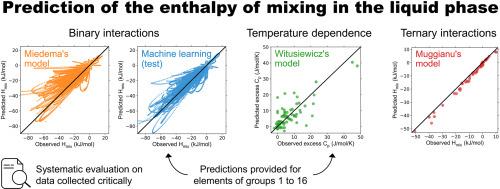
Data-driven study of the enthalpy of mixing in the liquid phase
The enthalpy of mixing in the liquid phase is a thermodynamic property reflecting interactions between elements that is key to predict phase transformations. Widely used models exist to predict it, but they have never been systematically evaluated. To address this, we collect a large amount of enthalpy of mixing data in binary liquids from a review of about 1000 thermodynamic evaluations. This allows us to clarify the prediction accuracy of Miedema's model which is state-of-the-art. We show that more accurate predictions can be obtained from a machine learning model based on LightGBM, and we provide them in 2415 binary systems. The data we collect also allows us to evaluate another empirical model to predict the excess heat capacity that we apply to 2211 binary liquids. We then extend the data collection to ternary metallic liquids and find that, when mixing is exothermic, extrapolations from the binary systems by Muggianu's model systematically lead to slight overestimations of roughly 10 % close to the equimolar composition. Therefore, our LightGBM model can provide reasonable estimates for ternary alloys and, by extension, for multicomponent alloys. Our findings extracted from rich datasets can be used to feed thermodynamic, empirical and machine learning models for material development. Our data, predictions, and code to generate machine learning descriptors from thermodynamic properties are all made available.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
4.00
自引率
16.70%
发文量
94
审稿时长
2.5 months
期刊介绍:
The design of industrial processes requires reliable thermodynamic data. CALPHAD (Computer Coupling of Phase Diagrams and Thermochemistry) aims to promote computational thermodynamics through development of models to represent thermodynamic properties for various phases which permit prediction of properties of multicomponent systems from those of binary and ternary subsystems, critical assessment of data and their incorporation into self-consistent databases, development of software to optimize and derive thermodynamic parameters and the development and use of databanks for calculations to improve understanding of various industrial and technological processes. This work is disseminated through the CALPHAD journal and its annual conference.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


