利用分子动力学模拟研究二氧化硅纳米颗粒存在时从水中溶剂萃取醋酸和丙酮的过程
IF 2.7
4区 生物学
Q2 BIOCHEMICAL RESEARCH METHODS
引用次数: 0
摘要
本研究采用分子动力学模拟预测醋酸和丙酮在水、甲苯和苯中的分子扩散系数。结果表明,COMPASS 是优化分子原子和结构的最佳力场,并将结果与实验方程进行了比较。研究了分子扩散系数在三种温度下的阿伦尼斯行为。使用甲苯和苯两种溶剂,在添加和不添加 SiO2 纳米粒子的情况下,研究了从水中萃取醋酸和丙酮的过程。绘制了不含和含二氧化硅纳米粒子的三种情况下的相对浓度变化图。为了定量检测结果,计算了萃取效率、选择性和分配系数。在没有二氧化硅纳米粒子的情况下,苯从水中萃取丙酮的效率为 65.748%,而在二氧化硅纳米粒子(浓度为 0.2231 wt%)存在的情况下,这一数值为 72.45%,这是因为纳米粒子的布朗运动增加了传质,从而提高了萃取效率。随着纳米颗粒浓度进一步增加到 1.7573 wt%,萃取效率下降到 61.276 %,这可能是由于二氧化硅纳米颗粒的积累和纳米颗粒自由运动的减少。本文章由计算机程序翻译,如有差异,请以英文原文为准。
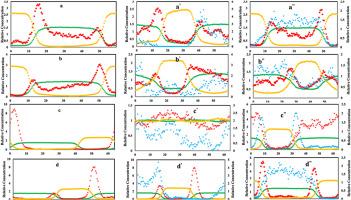
Investigation of solvent extraction of acetic acid and acetone from water in the presence of SiO2 nanoparticles using molecular dynamics simulation
In this study, molecular dynamics simulation was used to predict the molecular diffusion coefficient of acetic acid and acetone in water, toluene, and benzene. The results showed that COMPASS was the best force field to optimize the atoms and structure of molecules, and the results were compared with experimental equations. The Arrhenius behavior of the molecular diffusion coefficient was investigated at three temperatures. The extraction of acetic acid and acetone from water was investigated using two solvents, toluene, and benzene, with and without SiO2 nanoparticles. The relative concentration change diagram was drawn for three cases without and with SiO2 nanoparticles. To quantitatively examine the results, extraction efficiency, selectivity, and distribution coefficient were calculated. The extraction efficiency of acetone from water by benzene in the absence of silica nanoparticles was 65.748 %, this value in the presence of SiO2 nanoparticles with a concentration of 0.2231 wt% was 72.45 % due to the Brownian motion of the nanoparticles, which increased the mass transfer and as a result, the extraction efficiency. With the further increase of nanoparticles up to 1.7573 wt%, the extraction efficiency decreased to 61.276 %, which can be attributed to the accumulation of silica nanoparticles and the decrease in the free movement of nanoparticles.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of molecular graphics & modelling
生物-计算机:跨学科应用
CiteScore
5.50
自引率
6.90%
发文量
216
审稿时长
35 days
期刊介绍:
The Journal of Molecular Graphics and Modelling is devoted to the publication of papers on the uses of computers in theoretical investigations of molecular structure, function, interaction, and design. The scope of the journal includes all aspects of molecular modeling and computational chemistry, including, for instance, the study of molecular shape and properties, molecular simulations, protein and polymer engineering, drug design, materials design, structure-activity and structure-property relationships, database mining, and compound library design.
As a primary research journal, JMGM seeks to bring new knowledge to the attention of our readers. As such, submissions to the journal need to not only report results, but must draw conclusions and explore implications of the work presented. Authors are strongly encouraged to bear this in mind when preparing manuscripts. Routine applications of standard modelling approaches, providing only very limited new scientific insight, will not meet our criteria for publication. Reproducibility of reported calculations is an important issue. Wherever possible, we urge authors to enhance their papers with Supplementary Data, for example, in QSAR studies machine-readable versions of molecular datasets or in the development of new force-field parameters versions of the topology and force field parameter files. Routine applications of existing methods that do not lead to genuinely new insight will not be considered.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


