乙醇与硫代乙酰胺的酒精性肝病小鼠模型确定了作为人类疾病靶点的肝脏通路。
IF 3.7
3区 医学
Q2 GASTROENTEROLOGY & HEPATOLOGY
引用次数: 0
摘要
引言和目的:已知酒精相关性肝炎(AAH)会引起肝脏蛋白质组和肠道微生物群的改变。目前的动物模型很少模拟人类 AAH。我们的目标是建立一个与人类 AAH 非常相似的小鼠模型:雄性 C57BL/6N 小鼠被配对喂食对照组/增量乙醇 Lieber-DeCarli 日粮和硫代乙酰胺(TAA)12 周,以诱导 AAH。使用 LC-MS/MS 分析肝脏蛋白质组。利用 16s-rRNA 测序确定肠道细菌:结果:暴露于 EtOH+TAA 的小鼠肝脏甘油三酯(1.5 倍,p = 0.001)、促炎性(IL6,1.5 倍,p = 0.002 和 TNFα,1.7 倍,p = 0.01)、纤维化(TGF-β,2.7 倍,p = 0.01 和 Col1α1,2 倍,p = 0.01)和氧化标志物(GSH 和 SOD(-1.5 倍,p = 0.004 和 0.005))。EtOH+TAA肝脏的组织学表现为肝纤维化、脂肪变性和中性粒细胞浸润,与人类AAH相似。在 12 周的 EtOH+TAA 组中,脱硫杆菌、弯曲杆菌和棒状杆菌增加了 2 倍(p = 0.02)。EtOH+TAA处理的通路综合得分(CS,log10)显示,肝脏乙醇氧化(CS=1.93)、脂肪酸生物合成(CS=2.48)、坏死(CS=1.59)、胶原形成(CS=1.28)和缺氧(CS=0.68),并下调脂肪酸β-氧化(CS=2.37)、PPAR 信号转导(CS=1.35)、脂肪酸降解(CS=2.35)、胆汁酸代谢(CS=1.87)和氧化磷酸化(CS=1.50),正如在人类疾病中观察到的那样:结论:与之前已知的模型相比,在小鼠体内使用乙醇-硫代乙酰胺组合可更快地形成伴有纤维化的 AAH。差异蛋白表达与人类 AAH 中发现的改变途径密切相关,因此该模型比其他已知模型更能模拟人类疾病。硫代乙酰胺(TAA)可增强肝纤维化并模拟人类 AAH。本文章由计算机程序翻译,如有差异,请以英文原文为准。
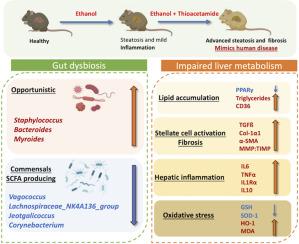
Ethanol with thioacetamide murine model of alcoholic liver disease identifies hepatic pathways as targets for the human disease
Introduction and Objectives
Hepatic proteome and gut microbiota alterations are known in alcohol-associated hepatitis (AAH). Current animal models sparsely mimic human AAH. We aimed to develop an murine model that closely resembled human AAH.
Materials and Methods
Male C57BL/6N mice were pair-fed control/incremental ethanol Lieber-DeCarli diets and thioacetamide (TAA) for 12-weeks to induce AAH. Hepatic proteome was analyzed using LC-MS/MS. Gut-bacteria was determined using 16s-rRNA sequencing.
Results
Mice exposed to EtOH+TAA displayed higher expression of liver triglycerides (1.5-fold, p = 0.001), pro-inflammatory (IL6, 1.5-fold, p = 0.002 and TNFα, 1.7-fold, p = 0.01), fibrotic (TGF-β, 2.7-fold, p = 0.01 and Col1α1, 2-fold, p = 0.01) and oxidative markers (GSH and SOD (-1.5 fold, p = 0.004 & 0.005 respectively)) as compared to EtOH alone. Histology of EtOH+TAA liver displayed pericellular liver fibrosis, increased steatosis, and neutrophil infiltration, which resembled human AAH. In the 12wk EtOH+TAA group, Desulfobacteria, Campylobacteria, and Patescibacteria increased by 2-fold (p = 0.02). Pathway combined score (CS, log10) in EtOH+TAA treatment showed upregulated hepatic ethanol oxidation (CS=1.93), fatty acid biosynthesis (CS=2.48), necrosis (CS=1.59), collagen formation (CS=1.28) and hypoxia (CS=0.68) and downregulated fatty acid beta-oxidation (CS=2.37), PPAR signaling (CS=1.35) fatty acid degradation (CS=2.35), bile acid metabolism (CS=1.87), and oxidative phosphorylation (CS=1.50), as observed in human disease.
Conclusions
Using an ethanol-thioacetamide combination in mice results in a faster establishment of AAH with fibrosis than previously known models. Differential protein expression strongly correlates with pathways found altered in human AAH, thus making the model mimic human disease better than other known models., respectively. Thioacetamide (TAA) was administered to enhance liver fibrosis and mimic human AAH.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Annals of hepatology
医学-胃肠肝病学
CiteScore
7.90
自引率
2.60%
发文量
183
审稿时长
4-8 weeks
期刊介绍:
Annals of Hepatology publishes original research on the biology and diseases of the liver in both humans and experimental models. Contributions may be submitted as regular articles. The journal also publishes concise reviews of both basic and clinical topics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


