作为麻醉药物的水溶性异丙酚衍生物的设计、合成和活性评估。
IF 2.5
4区 医学
Q3 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
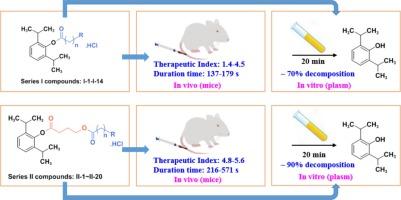
本研究以丙泊酚为先导化合物,根据其结构设计并合成了两个系列的水溶性衍生物。此外,还对合成化合物对小鼠的体内麻醉活性进行了评价,并测定了五个目标化合物的体外异丙酚释放率。研究结果表明,具有异丙酚+γ-羟丁酸+α-氨基乙酸或γ-氨基丁酸结构特征的系列 II 化合物比具有异丙酚+α-氨基乙酸或β-氨基丙酸结构特征的系列 I 化合物具有更高的治疗指数。此外,系列 II 化合物释放异丙酚的速率明显优于系列 I 化合物。在系列 II 化合物中,化合物 II-20 的治疗指数为 5.6(异丙酚 = 2.7),持续时间为 571 秒(异丙酚 = 57 秒),且在体内未观察到明显毒性,因此具有进一步开发的价值。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Design, synthesis, and activity evaluation of water-soluble propofol derivatives as anesthetic drugs
In this work, two series of water-soluble derivatives were designed and synthesized based on the structure of propofol as the lead compound. Furthermore, the anesthetic activities of the synthesized compounds were evaluated in vivo against mice, and the in vitro propofol release rate from five target compounds was determined. The findings of this study have shown that series II compounds which possess the structure feature of propofol + γ-hydroxybutyric acid + α-aminoacetate or γ-aminobutyrate have higher therapeutic index than that of series I compounds which possess the structure feature of propofol + α-aminoacetate or β-aminopropionate. In addition, the rate of propofol released from series II compounds was significantly better than that of series I compounds. Among series II compounds, compound II-20 had a therapeutic index of 5.6 (propofol = 2.7), a duration time of 571 s (propofol = 57 s), and no significant toxicity was observed in vivo, which made it valuable for further development.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
5.70
自引率
3.70%
发文量
463
审稿时长
27 days
期刊介绍:
Bioorganic & Medicinal Chemistry Letters presents preliminary experimental or theoretical research results of outstanding significance and timeliness on all aspects of science at the interface of chemistry and biology and on major advances in drug design and development. The journal publishes articles in the form of communications reporting experimental or theoretical results of special interest, and strives to provide maximum dissemination to a large, international audience.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


