由单一 AAV9 载体传递的 CRISPR-Cas9 介导的基因组编辑可抑制潜伏兔角膜炎模型中的 HSV-1 再激活
IF 4.7
2区 医学
Q2 MEDICINE, RESEARCH & EXPERIMENTAL
Molecular Therapy-Methods & Clinical Development
Pub Date : 2024-08-14
DOI:10.1016/j.omtm.2024.101303
引用次数: 0
摘要
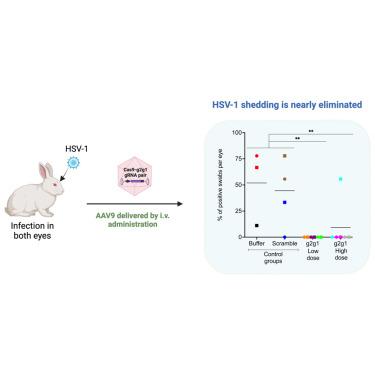
单纯疱疹病毒 1(HSV-1)角膜炎是全球失明的主要原因。在原发性感染期间,HSV-1 会进入三叉神经节,并形成终生潜伏期。虽然一些治疗方法可以减轻症状的严重程度并减少复发,但目前还没有治愈 HSV-1 角膜炎的方法。我们使用 CRISPR-Cas9 共同靶向编码两种重要 HSV-1 蛋白 ICP0 和 ICP27 的基因序列,作为治疗 HSV-1 角膜炎的一种潜在疗法。在HSV-1感染的Vero细胞中,通过质粒转染或AAV2载体转染表达Cas9核酸酶(SaCas9)和成对的引导RNA(gRNA),HSV-1病毒载量和滴度显著降低。脱靶评估显示,所选 gRNA 的脱靶编辑活性极低。随后,我们在HSV-1角膜炎潜伏兔模型中测试了我们的CRISPR-Cas9基因编辑方法。使用AAV8(Y733F)-SaCas9或AAV9-SaCas9一体化载体进行角膜瘢痕处理,病毒脱落率降低了50%以上。有趣的是,静脉注射相同的 AAV9-SaCas9 载体可消除 92% 治疗过的眼睛的病毒脱落。此外,经处理的三叉神经节显示 HSV-1 DNA 和 RNA 表达减少。我们的研究结果表明,单剂量 AAV9 一体式 CRISPR-Cas9 基因编辑是治疗 HSV-1 角膜炎的一种安全有效的策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
CRISPR-Cas9-mediated genome editing delivered by a single AAV9 vector inhibits HSV-1 reactivation in a latent rabbit keratitis model
Herpes simples virus 1 (HSV-1) keratitis is a major cause of blindness globally. During primary infection, HSV-1 travels to the trigeminal ganglia and establishes lifelong latency. Although some treatments can reduce symptom severity and recurrence, there is no cure for HSV-1 keratitis. We used CRISPR-Cas9 to co-target gene sequences encoding two essential HSV-1 proteins, ICP0 and ICP27, as a potential therapy for HSV-1 keratitis. In HSV-1-infected Vero cells, the HSV-1 viral load and titer were significantly reduced by plasmid transfection or AAV2 vector transduction expressing Cas9 nuclease from (SaCas9) and paired guide RNAs (gRNAs). Off-target assessment showed minimal off-target editing activity from the selected gRNAs. We then tested our CRISPR-Cas9 gene editing approach in a latent rabbit model of HSV-1 keratitis. Corneal scarification with all-in-one AAV8(Y733F)-SaCas9 or AAV9-SaCas9 vector reduced viral shedding by over 50%. Interestingly, intravenous administration of the same AAV9-SaCas9 vector eliminated viral shedding in 92% of treated eyes. In addition, treated trigeminal ganglia showed a reduction in HSV-1 DNA and RNA expression. Our results support the utility of single-dose AAV9 all-in-one CRISPR-Cas9 gene editing as a safe and effective strategy for treating HSV-1 keratitis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Molecular Therapy-Methods & Clinical Development
Biochemistry, Genetics and Molecular Biology-Molecular Biology
CiteScore
9.90
自引率
4.30%
发文量
163
审稿时长
12 weeks
期刊介绍:
The aim of Molecular Therapy—Methods & Clinical Development is to build upon the success of Molecular Therapy in publishing important peer-reviewed methods and procedures, as well as translational advances in the broad array of fields under the molecular therapy umbrella.
Topics of particular interest within the journal''s scope include:
Gene vector engineering and production,
Methods for targeted genome editing and engineering,
Methods and technology development for cell reprogramming and directed differentiation of pluripotent cells,
Methods for gene and cell vector delivery,
Development of biomaterials and nanoparticles for applications in gene and cell therapy and regenerative medicine,
Analysis of gene and cell vector biodistribution and tracking,
Pharmacology/toxicology studies of new and next-generation vectors,
Methods for cell isolation, engineering, culture, expansion, and transplantation,
Cell processing, storage, and banking for therapeutic application,
Preclinical and QC/QA assay development,
Translational and clinical scale-up and Good Manufacturing procedures and process development,
Clinical protocol development,
Computational and bioinformatic methods for analysis, modeling, or visualization of biological data,
Negotiating the regulatory approval process and obtaining such approval for clinical trials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


