隔离八面体铂诱导电子转移到超低含量钌掺杂尖晶石 Co3O4 以增强酸性整体水分离效果
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
开发一种高活性、高稳定性的氧进化反应(OER)电催化剂是利用可再生电力通过质子交换膜电解水(PEMWE)实现可持续高效制氢的理想选择,但这一研究仍具有挑战性。在此,我们报告了一种具有超低贵金属负载的强效铂/钌掺杂尖晶石氧化钴(PtRu-Co3O4)电催化剂,可用于酸性整体水分离。PtRu-Co3O4 表现出卓越的催化活性(100 mA cm-2 时为 1.63 V)和出色的稳定性,在 100 小时的运行过程中不会出现明显的性能衰减。实验分析和理论计算表明,掺杂铂能诱导电子转移到掺Ru的Co3O4上,优化氧中间产物的吸收能量,稳定金属氧键,从而通过吸附剂进化机制提高催化性能。因此,采用铂钌钴氧化物(PtRu-Co3O4)催化剂的 PEM 电解槽在 1.83 V 电压下可驱动 1.0 A cm-2 的电流密度,贵金属含量低的非铱基催化剂在制氢领域的应用前景广阔。本文章由计算机程序翻译,如有差异,请以英文原文为准。
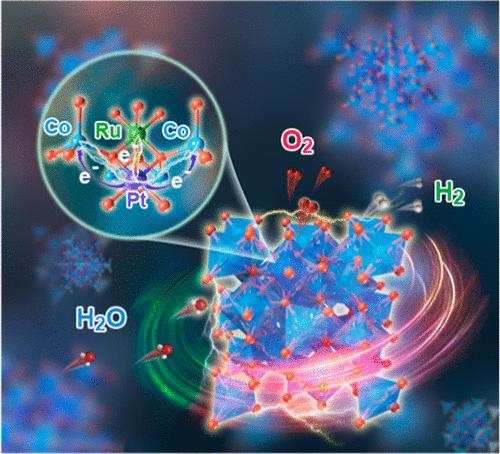
Isolated Octahedral Pt-Induced Electron Transfer to Ultralow-Content Ruthenium-Doped Spinel Co3O4 for Enhanced Acidic Overall Water Splitting
The development of a highly active and stable oxygen evolution reaction (OER) electrocatalyst is desirable for sustainable and efficient hydrogen production via proton exchange membrane water electrolysis (PEMWE) powered by renewable electricity yet challenging. Herein, we report a robust Pt/Ru-codoped spinel cobalt oxide (PtRu–Co3O4) electrocatalyst with an ultralow precious metal loading for acidic overall water splitting. PtRu–Co3O4 exhibits excellent catalytic activity (1.63 V at 100 mA cm–2) and outstanding stability without significant performance degradation for 100 h operation. Experimental analysis and theoretical calculations indicate that Pt doping can induce electron transfer to Ru-doped Co3O4, optimize the absorption energy of oxygen intermediates, and stabilize metal–oxygen bonds, thus enhancing the catalytic performance through an adsorbate-evolving mechanism. As a consequence, the PEM electrolyzer featuring PtRu–Co3O4 catalyst with low precious metal mass loading of 0.23 mg cm–2 can drive a current density of 1.0 A cm–2 at 1.83 V, revealing great promise for the application of noniridium-based catalysts with low contents of precious metal for hydrogen production.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


