阿米万他单抗对野生型表皮生长因子受体非小细胞肺癌肿瘤的疗效与配体表达水平相关。
IF 6.8
1区 医学
Q1 ONCOLOGY
引用次数: 0
摘要
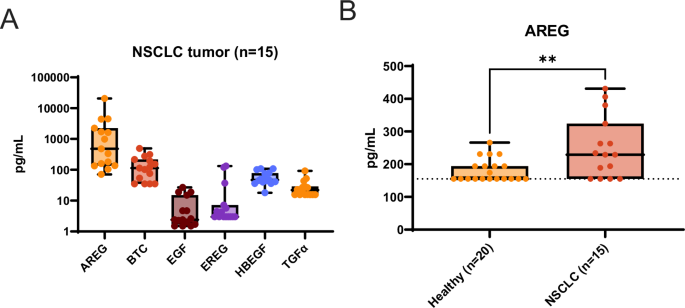
Amivantamab是美国食品及药物管理局(FDA)批准的一种靶向表皮生长因子受体(EGF)和金属受体(Met)的双特异性抗体,对表皮生长因子受体突变的非小细胞肺癌(NSCLC)具有临床活性。Amivantamab 的疗效已被证实与三种作用机制(MOA)有关:免疫细胞介导的杀伤、受体内化和降解以及抑制配体与表皮生长因子受体和 Met 受体的结合。在表皮生长因子受体配体中,我们发现安非拉酮(AREG)在野生型(WT)表皮生长因子受体(EGFRWT)NSCLC 原发性肿瘤中高度表达,NSCLC 患者的循环蛋白水平明显高于健康志愿者。用阿米万他单抗或 AREG 靶向抗体处理 AREG 刺激的 EGFRWT 细胞/肿瘤,可抑制配体诱导的信号传导和细胞/肿瘤的增殖/生长。在11种表皮生长因子受体WT NSCLC患者衍生异种移植模型中,amivantamab的疗效与AREG RNA水平相关。有趣的是,在这些模型中,阿米万他单抗的抗肿瘤活性与免疫细胞的 Fc 参与无关,这表明在这种情况下,配体阻断功能足以使阿米万他单抗发挥最大疗效。最后,我们证明在肺腺癌患者中,AREG 的高表达与表皮生长因子受体突变是相互排斥的。总之,这些数据:1)强调了表皮生长因子受体配体 AREG 是某些表皮生长因子受体突变型 NSCLC 模型中肿瘤生长的驱动因素;2)说明了阿米万他单抗在配体驱动的表皮生长因子受体突变型 NSCLC 中的临床前疗效;3)确定了 AREG 是表皮生长因子受体突变型 NSCLC 中阿米万他单抗活性的潜在预测性生物标志物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Amivantamab efficacy in wild-type EGFR NSCLC tumors correlates with levels of ligand expression
Amivantamab is an FDA-approved bispecific antibody targeting EGF and Met receptors, with clinical activity against EGFR mutant non-small cell lung cancer (NSCLC). Amivantamab efficacy has been demonstrated to be linked to three mechanisms of action (MOA): immune cell-mediated killing, receptor internalization and degradation, and inhibition of ligand binding to both EGFR and Met receptors. Among the EGFR ligands, we demonstrated that amphiregulin (AREG) is highly expressed in wild-type (WT) EGFR (EGFRWT) NSCLC primary tumors, with significantly higher circulating protein levels in NSCLC patients than in healthy volunteers. Treatment of AREG-stimulated EGFRWT cells/tumors with amivantamab or with an AREG-targeting antibody inhibited ligand-induced signaling and cell/tumor proliferation/growth. Across 11 EGFRWT NSCLC patient-derived xenograft models, amivantamab efficacy correlated with AREG RNA levels. Interestingly, in these models, amivantamab anti-tumor activity was independent of Fc engagement with immune cells, suggesting that, in this context, the ligand-blocking function is sufficient for amivantamab maximal efficacy. Finally, we demonstrated that in lung adenocarcinoma patients, high expression of AREG and EGFR mutations were mutually exclusive. In conclusion, these data 1) highlight EGFR ligand AREG as a driver of tumor growth in some EGFRWT NSCLC models, 2) illustrate the preclinical efficacy of amivantamab in ligand-driven EGFRWT NSCLC, and 3) identify AREG as a potential predictive biomarker for amivantamab activity in EGFRWT NSCLC.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

NPJ Precision Oncology
ONCOLOGY-
CiteScore
9.90
自引率
1.30%
发文量
87
审稿时长
18 weeks
期刊介绍:
Online-only and open access, npj Precision Oncology is an international, peer-reviewed journal dedicated to showcasing cutting-edge scientific research in all facets of precision oncology, spanning from fundamental science to translational applications and clinical medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


