为阿尔茨海默病研究开发远程测量技术的监管考虑因素
IF 12.4
1区 医学
Q1 HEALTH CARE SCIENCES & SERVICES
引用次数: 0
摘要
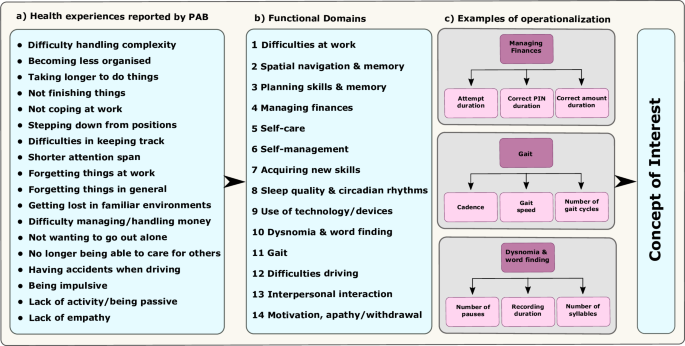
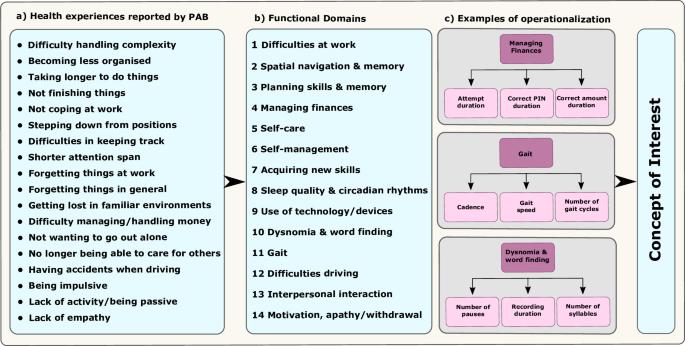
疾病和复发远程评估--阿尔茨海默病(RADAR-AD)联盟评估了用于评估阿尔茨海默病功能状态的远程测量技术(RMT)。该联盟与欧洲药品管理局 (EMA) 合作,就有意义的功能领域的确定、RMT 的选择和临床研究设计等方面获得反馈,以评估在 AD 临床研究中使用 RMT 的可行性。我们总结了反馈意见和经验教训,以指导未来的项目。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Regulatory considerations for developing remote measurement technologies for Alzheimer’s disease research
The Remote Assessment of Disease and Relapse – Alzheimer’s Disease (RADAR-AD) consortium evaluated remote measurement technologies (RMTs) for assessing functional status in AD. The consortium engaged with the European Medicines Agency (EMA) to obtain feedback on identification of meaningful functional domains, selection of RMTs and clinical study design to assess the feasibility of using RMTs in AD clinical studies. We summarized the feedback and the lessons learned to guide future projects.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

NPJ Digital Medicine
Multiple-
CiteScore
25.10
自引率
3.30%
发文量
170
审稿时长
15 weeks
期刊介绍:
npj Digital Medicine is an online open-access journal that focuses on publishing peer-reviewed research in the field of digital medicine. The journal covers various aspects of digital medicine, including the application and implementation of digital and mobile technologies in clinical settings, virtual healthcare, and the use of artificial intelligence and informatics.
The primary goal of the journal is to support innovation and the advancement of healthcare through the integration of new digital and mobile technologies. When determining if a manuscript is suitable for publication, the journal considers four important criteria: novelty, clinical relevance, scientific rigor, and digital innovation.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


