实现具有可调长度/组成和更强光催化活性的供体-受体纳米纤维的多功能战略
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
活结晶驱动自组装(CDSA)已成为以可控方式生成π共轭聚合物纳米纤维(CPNFs)的有效策略。然而,关于供体-受体(D-A)异质结 CPNF 的报道却极为罕见。由于缺乏 D-A π 共轭单元的合理设计准则,制备这些材料仍然是一项挑战。在本文中,我们报告了一种基于精心设计的 D-A 共轭高分子的多功能 CDSA 策略,其中缺电子的苯并噻二唑(BT)或二苯并[b,d]噻吩 5,5-二氧化物(FSO)单元被连接到低聚(对苯基乙炔)七聚物[BT-OPE7-BT,FSO-OPE7-FSO]的两端。这种低聚物两端的缺电子基团排列增强了 A-D-A π 共轭结构的堆叠相互作用。相反,在一串 OPE 单元中间有一个 BT 的 D-A-D 结构则会破坏堆积。我们采用带有末端炔烃的低聚物合成了二嵌段共聚物 BT-OPE7-BT-b-P2VP 和 BT-OPE7-BT-b-PNIPAM(P2VP = 聚(2-乙烯基吡啶),PNIPAM = 聚(N-异丙基丙烯酰胺))以及 FSO-OPE7-FSO-b-P2VP 和 FSO-OPE7-FSO-b-PNIPAM。用这些共聚物在乙醇中进行 CDSA 实验,能够通过自播种和播种生长生成长度可控的 CPNF,以及长度和成分可精确调节的嵌段漫游纤维。此外,以 BT-OPE7-BT 为核心的 D-A CPNF 在将硫化物光氧化成亚砜和将苄胺光氧化成 N-亚苄基苄胺方面具有光催化活性。考虑到所研究的低聚物成分范围和所形成的结构范围,我们认为,使用基于 D-A 的共低聚物的活 CDSA 策略为未来创造具有可编程结构以及多种功能和应用的 D-A CPNF 带来了机遇。本文章由计算机程序翻译,如有差异,请以英文原文为准。
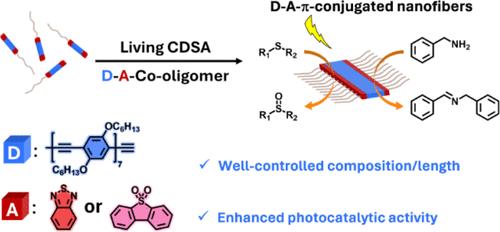
A Versatile Strategy toward Donor–Acceptor Nanofibers with Tunable Length/Composition and Enhanced Photocatalytic Activity
Living crystallization-driven self-assembly (CDSA) has emerged as an efficient strategy to generate nanofibers of π-conjugated polymers (CPNFs) in a controlled fashion. However, reports of donor–acceptor (D–A) heterojunction CPNFs are extremely rare. The preparation of these materials remains a challenge due to the lack of rational design guidelines for the D–A π-conjugated units. Herein, we report a versatile CDSA strategy based upon carefully designed D–A-co-oligomers in which electron-deficient benzothiadiazole (BT) or dibenzo[b,d]thiophene 5,5-dioxide (FSO) units are attached to the two ends of an oligo(p-phenylene ethynylene) heptamer [BT-OPE7-BT, FSO-OPE7-FSO]. This arrangement with the electron-deficient groups at the two ends of the oligomer enhances the stacking interaction of the A–D–A π-conjugated structure. In contrast, D–A–D structures with a single BT in the middle of a string of OPE units disrupt the packing. We employed oligomers with a terminal alkyne to synthesize diblock copolymers BT-OPE7-BT-b-P2VP and BT-OPE7-BT-b-PNIPAM (P2VP = poly(2-vinylpyridine), PNIPAM = poly(N-isopropylacrylamide)) and FSO-OPE7-FSO-b-P2VP and FSO-OPE7-FSO-b-PNIPAM. CDSA experiments with these copolymers in ethanol were able to generate CPNFs of controlled length by both self-seeding and seeded growth as well as block comicelles with precisely tunable length and composition. Furthermore, the D–A CPNFs with a BT-OPE7-BT-based core demonstrate photocatalytic activity for the photooxidation of sulfide to sulfoxide and benzylamine to N-benzylidenebenzylamine. Given the scope of the oligomer compositions examined and the range of structures formed, we believe that the living CDSA strategy with D–A-based co-oligomers opens future opportunities for the creation of D–A CPNFs with programmable architectures as well as diverse functionalities and applications.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


