癌症原发组织制约转移灶的生长和新陈代谢
IF 18.9
1区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
摘要
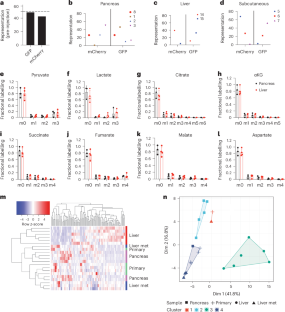
转移瘤由原发肿瘤扩散的癌细胞亚群产生1,2。癌细胞在新的组织部位生长的能力受遗传和表观遗传变化的影响,这些变化对疾病的发生和发展非常重要,但仅凭这些因素并不能预测癌症是否转移以及转移到何处3,4。特定癌症类型会转移到一致的组织亚群,这表明原发肿瘤相关因素会影响癌症的生长位置。我们发现原发性胰腺肿瘤和转移性胰腺肿瘤在新陈代谢方面有相似之处,而且原发性胰腺肿瘤和转移性胰腺肿瘤细胞的肿瘤诱发能力和增殖能力在原发部位比在转移部位更强。此外,将细胞作为肿瘤在肺部或肝部繁殖不会增强它们在这些部位形成大肿瘤的相对能力,不会改变它们在原发部位生长的偏好,也不会稳定地改变它们相对于原发肿瘤的新陈代谢方面。原发性肝癌和肺癌细胞也表现出在原发部位生长的偏好,而不是转移部位。这些数据表明,原发肿瘤组织会影响原发和转移肿瘤的新陈代谢,并可能影响癌细胞的转移位置。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Cancer tissue of origin constrains the growth and metabolism of metastases
Metastases arise from subsets of cancer cells that disseminate from the primary tumour1,2. The ability of cancer cells to thrive in a new tissue site is influenced by genetic and epigenetic changes that are important for disease initiation and progression, but these factors alone do not predict if and where cancers metastasize3,4. Specific cancer types metastasize to consistent subsets of tissues, suggesting that primary tumour-associated factors influence where cancers can grow. We find primary and metastatic pancreatic tumours have metabolic similarities and that the tumour-initiating capacity and proliferation of both primary-derived and metastasis-derived cells is favoured in the primary site relative to the metastatic site. Moreover, propagating cells as tumours in the lung or the liver does not enhance their relative ability to form large tumours in those sites, change their preference to grow in the primary site, nor stably alter aspects of their metabolism relative to primary tumours. Primary liver and lung cancer cells also exhibit a preference to grow in their primary site relative to metastatic sites. These data suggest cancer tissue of origin influences both primary and metastatic tumour metabolism and may impact where cancer cells can metastasize. Sivanand et al. survey different types of cancers and study how the metabolic profile of the primary cancer site influences the metabolism of the metastatic cells, thus influencing sites of metastasis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature metabolism
ENDOCRINOLOGY & METABOLISM-
CiteScore
27.50
自引率
2.40%
发文量
170
期刊介绍:
Nature Metabolism is a peer-reviewed scientific journal that covers a broad range of topics in metabolism research. It aims to advance the understanding of metabolic and homeostatic processes at a cellular and physiological level. The journal publishes research from various fields, including fundamental cell biology, basic biomedical and translational research, and integrative physiology. It focuses on how cellular metabolism affects cellular function, the physiology and homeostasis of organs and tissues, and the regulation of organismal energy homeostasis. It also investigates the molecular pathophysiology of metabolic diseases such as diabetes and obesity, as well as their treatment. Nature Metabolism follows the standards of other Nature-branded journals, with a dedicated team of professional editors, rigorous peer-review process, high standards of copy-editing and production, swift publication, and editorial independence. The journal has a high impact factor, has a certain influence in the international area, and is deeply concerned and cited by the majority of scholars.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


