MSK1是经验和安巴碱依赖性增强空间参照记忆和逆转学习的必要条件。
IF 4.6
2区 医学
Q1 NEUROSCIENCES
引用次数: 0
摘要
人们对能改善各种认知模式和认知障碍的药理制剂--促智剂的开发产生了浓厚的兴趣。其中一类认知增强剂--安非他酮类药物--因能改善与神经发育、神经退行性和精神疾病动物模型相关的认知能力,以及与年龄相关的认知障碍,而引起了人们的特别关注。安帕金类药物能提高中枢神经系统的 BDNF 水平,人们认为正是通过这种提高,安帕金类药物才发挥了有益的作用。然而,是什么将 BDNF 的升高转化为持久的认知能力增强尚不清楚。我们之前已经证明,MSK1 凭借其调控基因转录的能力,将与环境富集相关的 BDNF 升高转化为分子、突触、认知和基因组适应性,这些适应性是富集诱导的突触可塑性增强以及学习和记忆增强的基础,MSK1 的这一特性在人的一生中都会保持。为了确定 MSK1 是否同样能将安帕金诱导的 BDNF 升高转化为认知增强,我们在雄性 WT 小鼠和 MSK1 激酶活性发生突变的雄性小鼠体内测试了安帕金(CX929)。我们发现,安非他明依赖性地改善空间参照记忆和认知灵活性,以及与安非他明和经验相关的 BDNF 和可塑性相关蛋白 Arc 的升高,都需要 MSK1。这些观察结果表明,MSK1是安非他明类药物对认知功能产生有益影响的关键因素,并进一步确定了MSK1是提高BDNF的促智策略的枢纽。本文章由计算机程序翻译,如有差异,请以英文原文为准。
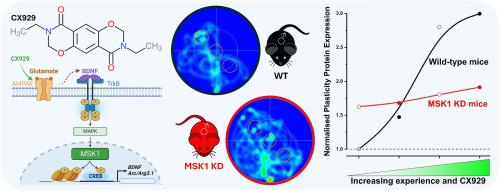
MSK1 is required for the experience- and ampakine-dependent enhancement of spatial reference memory and reversal learning and for the induction of Arc and BDNF
There is considerable interest in the development of nootropics, pharmacological agents that can improve cognition across a range of both cognitive modalities and cognitive disabilities. One class of cognitive enhancers, the ampakines, has attracted particular attention by virtue of improving cognition associated with animal models of neurodevelopmental, neurodegenerative, and psychiatric conditions, as well as in age-related cognitive impairment. Ampakines elevate CNS levels of BDNF, and it is through this elevation that their beneficial actions are believed to occur. However, what transduces the elevation of BDNF into long-lasting cognitive enhancement is not known. We have previously shown that MSK1, by virtue of its ability to regulate gene transcription, converts the elevation of BDNF associated with environmental enrichment into molecular, synaptic, cognitive and genomic adaptations that underlie enrichment-induced enhanced synaptic plasticity and learning and memory, a property that MSK1 retains across the lifespan. To establish whether MSK1 similarly converts ampakine-induced elevations of BDNF into cognitive enhancement we tested an ampakine (CX929) in male WT mice and in male mice in which the kinase activity of MSK1 was inactivated. We found that MSK1 is required for the ampakine-dependent improvement in spatial reference memory and cognitive flexibility, and for the elevations of BDNF and the plasticity-related protein Arc associated with ampakines and experience. These observations implicate MSK1 as a key enabler of the beneficial effects of ampakines on cognitive function, and furthermore identify MSK1 as a hub for BDNF-elevating nootropic strategies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Neuropharmacology
医学-神经科学
CiteScore
10.00
自引率
4.30%
发文量
288
审稿时长
45 days
期刊介绍:
Neuropharmacology publishes high quality, original research and review articles within the discipline of neuroscience, especially articles with a neuropharmacological component. However, papers within any area of neuroscience will be considered. The journal does not usually accept clinical research, although preclinical neuropharmacological studies in humans may be considered. The journal only considers submissions in which the chemical structures and compositions of experimental agents are readily available in the literature or disclosed by the authors in the submitted manuscript. Only in exceptional circumstances will natural products be considered, and then only if the preparation is well defined by scientific means. Neuropharmacology publishes articles of any length (original research and reviews).
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


