CircVPS8通过作为MKRN1、SOX15和HNF4A的支架,促进胶质瘤干细胞的恶性表型并抑制其铁细胞生成。
IF 6.9
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
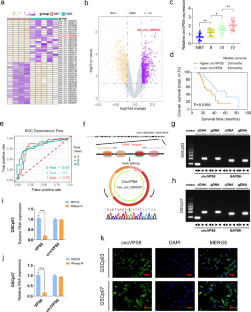
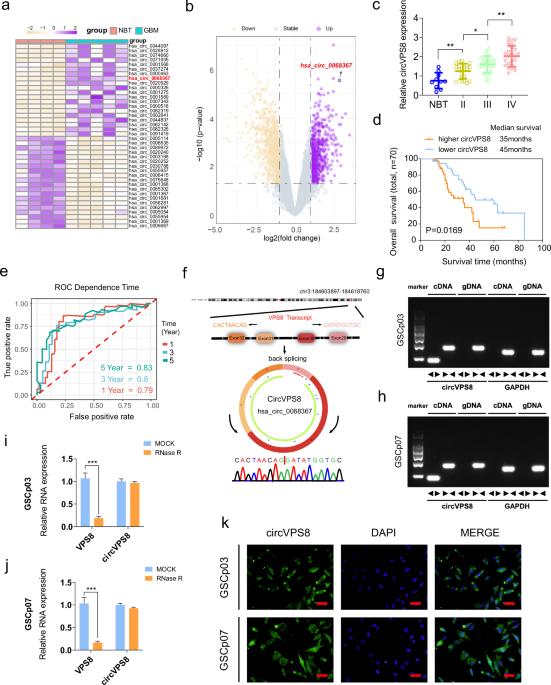
在胶质母细胞瘤领域,针对特定铁突变靶点的治疗干预取得了令人振奋的突破。然而,circRNA调控铁突变途径的确切机制尚未完全阐明。在这里,我们发现了一种在胶质母细胞瘤中高表达的新型 circRNA--circVPS8。我们的研究结果表明,circVPS8能增强胶质瘤干细胞的活力、增殖、成球能力和干性。此外,它还能抑制胶质细胞干细胞的铁突变。体内实验进一步证实了circVPS8对胶质母细胞瘤生长的促进作用。从机理上讲,circVPS8 可作为支架与 MKRN1 和 SOX15 结合,从而促进 MKRN1 的泛素化和 SOX15 的降解。由于竞争性结合,MKRN1 对 HNF4A 的泛素化能力降低,导致 HNF4A 表达升高。HNF4A 表达的增加与 SOX15 表达的减少可协同抑制胶质母细胞瘤中的铁突变。总之,我们的研究强调了circVPS8是一个有前景的治疗靶点,并为胶质母细胞瘤的临床靶向治疗提供了有价值的见解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
CircVPS8 promotes the malignant phenotype and inhibits ferroptosis of glioma stem cells by acting as a scaffold for MKRN1, SOX15 and HNF4A
Exciting breakthroughs have been achieved in the field of glioblastoma with therapeutic interventions targeting specific ferroptosis targets. Nonetheless, the precise mechanisms through which circRNAs regulate the ferroptosis pathway have yet to be fully elucidated. Here we have identified a novel circRNA, circVPS8, which is highly expressed in glioblastoma. Our findings demonstrated that circVPS8 enhances glioma stem cells’ viability, proliferation, sphere-forming ability, and stemness. Additionally, it inhibits ferroptosis in GSCs. In vivo, experiments further supported the promotion of glioblastoma growth by circVPS8. Mechanistically, circVPS8 acts as a scaffold, binding to both MKRN1 and SOX15, thus facilitating the ubiquitination of MKRN1 and subsequent degradation of SOX15. Due to competitive binding, the ubiquitination ability of MKRN1 towards HNF4A is reduced, leading to elevated HNF4A expression. Increased HNF4A expression, along with decreased SOX15 expression, synergistically inhibits ferroptosis in glioblastoma. Overall, our study highlights circVPS8 as a promising therapeutic target and provides valuable insights for clinically targeted therapy of glioblastoma.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Oncogene
医学-生化与分子生物学
CiteScore
15.30
自引率
1.20%
发文量
404
审稿时长
1 months
期刊介绍:
Oncogene is dedicated to advancing our understanding of cancer processes through the publication of exceptional research. The journal seeks to disseminate work that challenges conventional theories and contributes to establishing new paradigms in the etio-pathogenesis, diagnosis, treatment, or prevention of cancers. Emphasis is placed on research shedding light on processes driving metastatic spread and providing crucial insights into cancer biology beyond existing knowledge.
Areas covered include the cellular and molecular biology of cancer, resistance to cancer therapies, and the development of improved approaches to enhance survival. Oncogene spans the spectrum of cancer biology, from fundamental and theoretical work to translational, applied, and clinical research, including early and late Phase clinical trials, particularly those with biologic and translational endpoints.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


