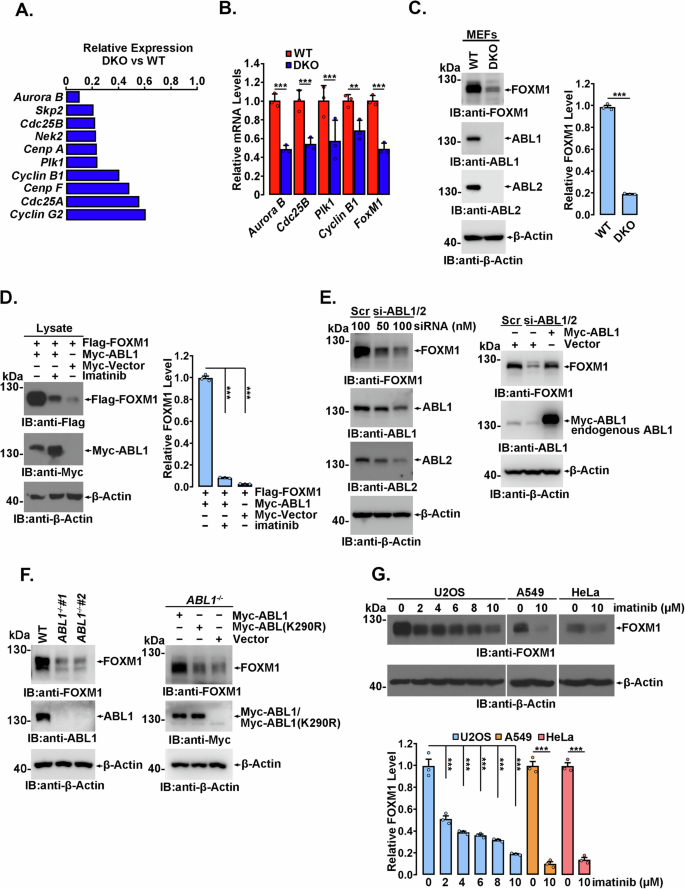
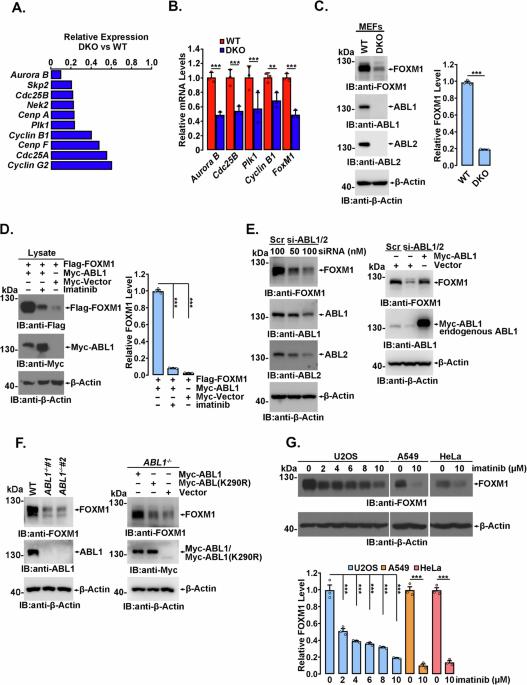
ABL1 介导的磷酸化通过增加 FOXM1 的稳定性促进 FOXM1 相关致瘤性。
IF 13.7
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
转录因子FOXM1在细胞周期进展和肿瘤发生中起着关键作用,在快速增殖的细胞和各种肿瘤组织中高表达,FOXM1的高表达与预后不良有关。然而,导致 FOXM1 失调的机制尚未完全明了。在这里,我们发现非受体酪氨酸激酶 ABL1 促进了 FOXM1 的高表达和 FOXM1 依赖性肿瘤的发展。从机制上讲,ABL1 直接与 FOXM1 结合,并在多个酪氨酸(Y)残基上介导 FOXM1 磷酸化。在这些磷酸化 Y 位点中,pY575 对 FOXM1 的稳定性不可或缺,因为该位点的磷酸化可保护 FOXM1 免受泛素-蛋白酶体降解。FOXM1 与 CDH1(E3 泛素连接酶 anaphase-promoting complex/cyclosome(APC/C)的辅助激活剂)的相互作用受到 Y575 磷酸化的显著抑制,而 CDH1 是负责 FOXM1 降解的 E3 泛素连接酶。磷酸化缺陷的 FOXM1(Y575F)突变体表现出泛素化增加、半衰期缩短,因此丰度大大降低。与野生型细胞相比,通过CRISPR/Cas9技术生成的表达内源性FOXM1(Y575F)的同源Cr-Y575F细胞系明显延迟了有丝分裂进程,阻碍了集落的形成,并抑制了异种移植肿瘤的生长。总之,我们的研究表明,ABL1激酶参与了FOXM1的高表达,为ABL1可能成为治疗FOXM1高表达肿瘤的靶点提供了明确证据。本文章由计算机程序翻译,如有差异,请以英文原文为准。
ABL1-mediated phosphorylation promotes FOXM1-related tumorigenicity by Increasing FOXM1 stability
The transcription factor FOXM1, which plays critical roles in cell cycle progression and tumorigenesis, is highly expressed in rapidly proliferating cells and various tumor tissues, and high FOXM1 expression is related to a poor prognosis. However, the mechanism responsible for FOXM1 dysregulation is not fully understood. Here, we show that ABL1, a nonreceptor tyrosine kinase, contributes to the high expression of FOXM1 and FOXM1-dependent tumor development. Mechanistically, ABL1 directly binds FOXM1 and mediates FOXM1 phosphorylation at multiple tyrosine (Y) residues. Among these phospho-Y sites, pY575 is indispensable for FOXM1 stability as phosphorylation at this site protects FOXM1 from ubiquitin-proteasomal degradation. The interaction of FOXM1 with CDH1, a coactivator of the E3 ubiquitin ligase anaphase-promoting complex/cyclosome (APC/C), which is responsible for FOXM1 degradation, is significantly inhibited by Y575 phosphorylation. The phospho-deficient FOXM1(Y575F) mutant exhibited increased ubiquitination, a shortened half-life, and consequently a substantially decreased abundance. Compared to wild-type cells, a homozygous Cr-Y575F cell line expressing endogenous FOXM1(Y575F) that was generated by CRISPR/Cas9 showed obviously delayed mitosis progression, impeded colony formation and inhibited xenotransplanted tumor growth. Overall, our study demonstrates that ABL1 kinase is involved in high FOXM1 expression, providing clear evidence that ABL1 may act as a therapeutic target for the treatment of tumors with high FOXM1 expression.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell Death and Differentiation
生物-生化与分子生物学
CiteScore
24.70
自引率
1.60%
发文量
181
审稿时长
3 months
期刊介绍:
Mission, vision and values of Cell Death & Differentiation:
To devote itself to scientific excellence in the field of cell biology, molecular biology, and biochemistry of cell death and disease.
To provide a unified forum for scientists and clinical researchers
It is committed to the rapid publication of high quality original papers relating to these subjects, together with topical, usually solicited, reviews, meeting reports, editorial correspondence and occasional commentaries on controversial and scientifically informative issues.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


