逆转录病毒复制载体介导的基因疗法与抗-PD-1 抗体相结合在小鼠胰腺癌模型中的治疗活性。
IF 4.8
3区 医学
Q1 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 0
摘要
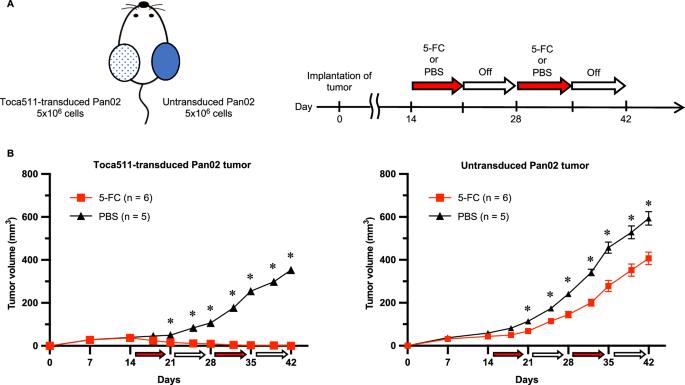
Toca 511 是一种编码酵母胞嘧啶脱氨酶(yCD)基因的肿瘤选择性逆转录病毒复制载体,通过瘤内原药 5-氟胞嘧啶(5-FC)在 yCD 的作用下转化为活性药物 5-氟尿嘧啶,从而直接发挥抗肿瘤作用。托卡 511/5-FC 治疗还可诱导抗肿瘤免疫。在这里,我们首先在免疫功能正常的小鼠胰腺癌模型中研究了 Toca 511/5-FC 治疗激活的抗肿瘤免疫反应。然后,我们评估了与抗程序性细胞死亡蛋白 1 抗体联合使用所取得的治疗效果。在双侧皮下肿瘤模型中,与对照组相比,在 Toca 511 未转导的对侧肿瘤中观察到 CD8+ T 细胞介导的细胞毒性增强和 T 细胞浸润增加。此外,在治疗过程中,CD8+ T 细胞上的 T 细胞协同抑制受体的表达水平也有所增加。在双侧皮下肿瘤模型中,联合疗法对肿瘤生长的抑制作用明显强于单一疗法。在正位肿瘤和腹膜播散模型中,联合疗法可使转导的正位肿瘤和未转导的腹膜播散肿瘤完全消退。因此,Toca 511/5-FC 治疗可诱导全身性抗肿瘤免疫反应,这种联合疗法可能是治疗转移性胰腺癌的一种有前途的临床策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Therapeutic activity of retroviral replicating vector-mediated gene therapy in combination with anti-PD-1 antibody in a murine pancreatic cancer model
Toca 511, a tumor-selective retroviral replicating vector encoding the yeast cytosine deaminase (yCD) gene, exerts direct antitumor effects through intratumoral prodrug 5-fluorocytosine (5-FC) conversion to active drug 5-fluorouracil by yCD, and has demonstrated therapeutic efficacy in preclinical and clinical trials of various cancers. Toca 511/5-FC treatment may also induce antitumor immunity. Here, we first examined antitumor immune responses activated by Toca 511/5-FC treatment in an immunocompetent murine pancreatic cancer model. We then evaluated the therapeutic effects achieved in combination with anti-programmed cell death protein 1 antibody. In the bilateral subcutaneous tumor model, as compared with the control group, enhanced CD8+ T-cell-mediated cytotoxicity and increased T-cell infiltration in Toca 511-untransduced contralateral tumors were observed. Furthermore, the expression levels of T-cell co-inhibitory receptors on CD8+ T-cells increased during treatment. In the bilateral subcutaneous tumor model, combination therapy showed significantly stronger tumor growth inhibition than that achieved with either monotherapy. In an orthotopic tumor and peritoneal dissemination model, the combination therapy resulted in complete regression in both transduced orthotopic tumors and untransduced peritoneal dissemination. Thus, Toca 511/5-FC treatment induced a systemic antitumor immune response, and the combination therapy could be a promising clinical strategy for treating metastatic pancreatic cancer.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cancer gene therapy
医学-生物工程与应用微生物
CiteScore
10.20
自引率
0.00%
发文量
150
审稿时长
4-8 weeks
期刊介绍:
Cancer Gene Therapy is the essential gene and cellular therapy resource for cancer researchers and clinicians, keeping readers up to date with the latest developments in gene and cellular therapies for cancer. The journal publishes original laboratory and clinical research papers, case reports and review articles. Publication topics include RNAi approaches, drug resistance, hematopoietic progenitor cell gene transfer, cancer stem cells, cellular therapies, homologous recombination, ribozyme technology, antisense technology, tumor immunotherapy and tumor suppressors, translational research, cancer therapy, gene delivery systems (viral and non-viral), anti-gene therapy (antisense, siRNA & ribozymes), apoptosis; mechanisms and therapies, vaccine development, immunology and immunotherapy, DNA synthesis and repair.
Cancer Gene Therapy publishes the results of laboratory investigations, preclinical studies, and clinical trials in the field of gene transfer/gene therapy and cellular therapies as applied to cancer research. Types of articles published include original research articles; case reports; brief communications; review articles in the main fields of drug resistance/sensitivity, gene therapy, cellular therapy, tumor suppressor and anti-oncogene therapy, cytokine/tumor immunotherapy, etc.; industry perspectives; and letters to the editor.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


