眼镜蛇毒液的分子剖析凸显了肝磷脂是一种治疗吐毒眼镜蛇中毒的解毒剂。
IF 15.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
摘要
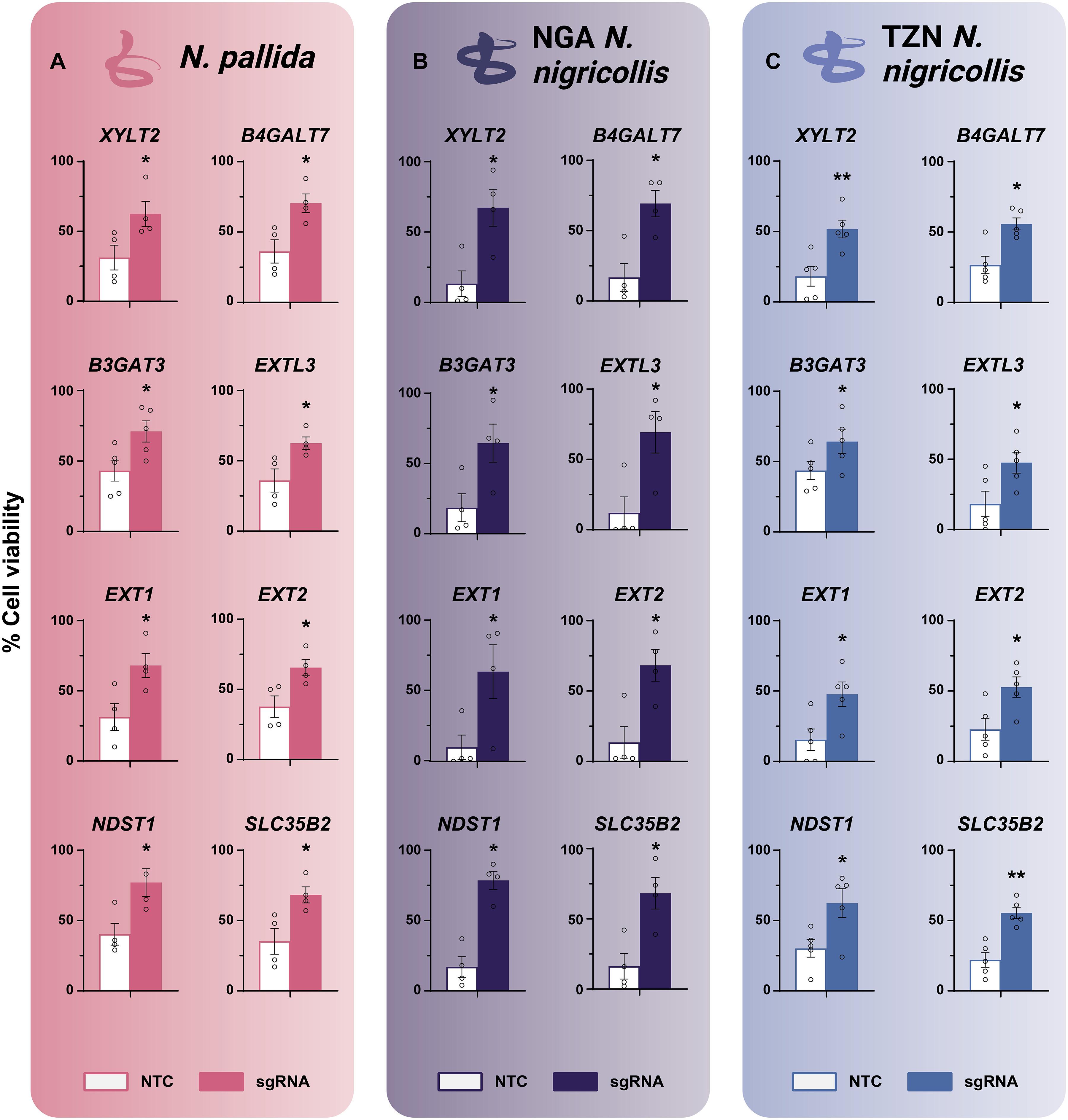
每年约有 180 万人被蛇咬伤。目前的治疗标准是使用基于抗体的抗蛇毒血清,但这种抗蛇毒血清很难获得,而且通常对局部组织损伤无效,而局部组织损伤是发病的主要原因。在这里,我们利用集合全基因组CRISPR基因敲除筛选来确定人类基因,这些基因一旦被锁定,就会改变细胞对喷吐眼镜蛇毒液的反应。我们发现,大部分对毒液细胞毒性具有抗性的修饰基因控制着蛋白多糖的生物合成,包括 EXT1、B4GALT7、EXT2、EXTL3、XYLT2、NDST1 和 SLC35B2,我们对这些基因进行了独立验证。这一发现表明肝磷脂可能是抑制剂。肝素类药物通过与三指细胞毒素结合来阻止毒液的细胞毒性,美国食品和药物管理局批准的肝素类药物锡扎肝素(tinzaparin)在通过医疗相关途径和剂量给药时可减少小鼠的组织损伤。总之,我们对眼镜蛇毒的细胞毒性进行了系统的分子剖析,为我们如何更好地治疗眼镜蛇蛇咬伤提供了启示。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Molecular dissection of cobra venom highlights heparinoids as an antidote for spitting cobra envenoming
Snakebites affect about 1.8 million people annually. The current standard of care involves antibody-based antivenoms, which can be difficult to access and are generally not effective against local tissue injury, the primary cause of morbidity. Here, we used a pooled whole-genome CRISPR knockout screen to define human genes that, when targeted, modify cell responses to spitting cobra venoms. A large portion of modifying genes that conferred resistance to venom cytotoxicity was found to control proteoglycan biosynthesis, including EXT1, B4GALT7, EXT2, EXTL3, XYLT2, NDST1, and SLC35B2, which we validated independently. This finding suggested heparinoids as possible inhibitors. Heparinoids prevented venom cytotoxicity through binding to three-finger cytotoxins, and the US Food and Drug Administration–approved heparinoid tinzaparin was found to reduce tissue damage in mice when given via a medically relevant route and dose. Overall, our systematic molecular dissection of cobra venom cytotoxicity provides insight into how we can better treat cobra snakebite envenoming.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Science Translational Medicine
CELL BIOLOGY-MEDICINE, RESEARCH & EXPERIMENTAL
CiteScore
26.70
自引率
1.20%
发文量
309
审稿时长
1.7 months
期刊介绍:
Science Translational Medicine is an online journal that focuses on publishing research at the intersection of science, engineering, and medicine. The goal of the journal is to promote human health by providing a platform for researchers from various disciplines to communicate their latest advancements in biomedical, translational, and clinical research.
The journal aims to address the slow translation of scientific knowledge into effective treatments and health measures. It publishes articles that fill the knowledge gaps between preclinical research and medical applications, with a focus on accelerating the translation of knowledge into new ways of preventing, diagnosing, and treating human diseases.
The scope of Science Translational Medicine includes various areas such as cardiovascular disease, immunology/vaccines, metabolism/diabetes/obesity, neuroscience/neurology/psychiatry, cancer, infectious diseases, policy, behavior, bioengineering, chemical genomics/drug discovery, imaging, applied physical sciences, medical nanotechnology, drug delivery, biomarkers, gene therapy/regenerative medicine, toxicology and pharmacokinetics, data mining, cell culture, animal and human studies, medical informatics, and other interdisciplinary approaches to medicine.
The target audience of the journal includes researchers and management in academia, government, and the biotechnology and pharmaceutical industries. It is also relevant to physician scientists, regulators, policy makers, investors, business developers, and funding agencies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


