作为碱性海水中氢气进化高效催化剂的镍的热诱导异构化和磷化
IF 8.6
2区 材料科学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
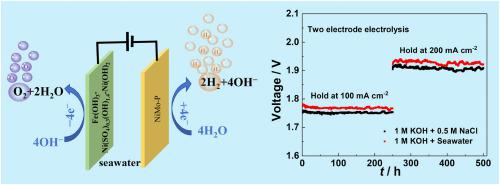
开发性能优异的电解海水催化剂对于高效制氢至关重要。本研究通过钼酸镍铵中镍的热诱导异构化和磷化,合成了锚定在镍钼氧化物上的镍珀纳米颗粒,简称为镍钼珀。物理化学特征显示,丰富的 NiP 纳米粒子均匀地分布在 NiMo 氧化物上。电化学数据显示,在碱性模拟海水中,仅 103 mV 的过电位就足以达到 -100 mA cm,明显低于铂箔(179 mV)和商用 Pt/C(165 mV)。根据密度泛函理论计算,NiMo-P 纳米粒子的水解离活性和氢解吸能力均优于铂,这可能是 NiMo-P 纳米粒子具有显著活性的原因。同时,NiMo-P 还具有出色的稳定性,这一点可以从计时器曲线中得到证明。在 500 小时后,电流仍保持在初始值的 98.1%,这归功于蚀刻-水解方法加强了催化剂与载体之间的相互作用。此外,阴极对氯离子的固有排斥性也有效地避免了化学腐蚀。重要的是,当与之前报告的阳极结合使用时,NiMo-P 还能在碱性海水中表现出卓越的性能。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Heat-induced aliquation and phosphating of nickel as efficient catalysts for hydrogen evolution in alkaline seawater
Developing electrolytic seawater catalysts with excellent performance is crucial for efficient hydrogen production. In this study, NiP nanoparticles anchored on NiMo oxides, denoted as NiMo-P, are synthesized through heat-induced aliquation and phosphating of Ni in the ammonium nickel molybdate. Physicochemical characterizations reveal that the abundant NiP nanoparticles are uniformly distributed on NiMo oxides. Electrochemical data reveal that a mere overpotential of 103 mV is sufficient to achieve −100 mA cm in alkaline simulated seawater, which is significantly lower than that of Pt foil (179 mV) and commercial Pt/C (165 mV). This remarkable activity observed in NiMo-P may be due to the superior water dissociation activity and hydrogen desorption ability of the NiP nanoparticles, as calculated by density functional theory, which surpasses that of Pt. Meanwhile, the NiMo-P exhibits outstanding stability, as evidenced by the chronoamperometric curve. The current remains at 98.1% of its initial value after 500 h, which can be attributed to the etching-hydrolysis method that strengthens the catalyst-carrier interaction. Besides, the inherent repulsion toward chlorine ions at the cathode effectively avoids chemical corrosion. Importantly, when coupled with the previously reported anode, NiMo-P also exhibits exceptional performance in alkaline seawater.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Materials Today Energy
Materials Science-Materials Science (miscellaneous)
CiteScore
15.10
自引率
7.50%
发文量
291
审稿时长
15 days
期刊介绍:
Materials Today Energy is a multi-disciplinary, rapid-publication journal focused on all aspects of materials for energy.
Materials Today Energy provides a forum for the discussion of high quality research that is helping define the inclusive, growing field of energy materials.
Part of the Materials Today family, Materials Today Energy offers authors rigorous peer review, rapid decisions, and high visibility. The editors welcome comprehensive articles, short communications and reviews on both theoretical and experimental work in relation to energy harvesting, conversion, storage and distribution, on topics including but not limited to:
-Solar energy conversion
-Hydrogen generation
-Photocatalysis
-Thermoelectric materials and devices
-Materials for nuclear energy applications
-Materials for Energy Storage
-Environment protection
-Sustainable and green materials
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


