通过 N-苄基活化策略对偶氮进行 Radical meta-C-H 卤化。
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
六元 N-teroarenes 的区域选择性卤化对于精确的功能衍生至关重要。我们介绍了一种利用 N-苄基活化策略,通过亲电卤素自由基加成法对吡啶、喹啉和异喹啉进行元选择性卤化的方法。该方法实现了吡啶中的 C3- 和 C5-二卤化、喹啉中的 C3- 和 C6-二卤化以及异喹啉中的 C3-单卤化。通过放大反应和喹啉衍生物与生物分子片段的溴化反应,验证了该方法的可行性和潜在应用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
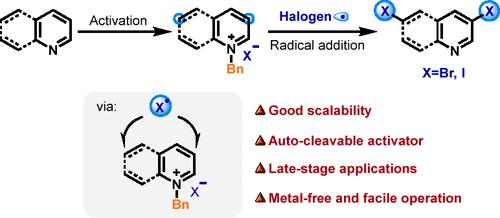
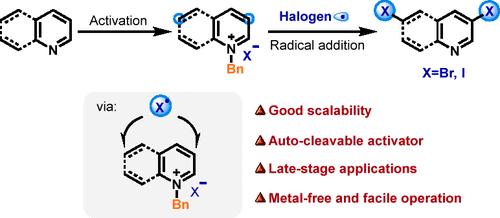
Radical meta-C–H Halogenation of Azines via N-Benzyl Activation Strategy
Regioselective halogenation of six-membered N-heteroarenes is crucial for precise functional derivatization. We present a meta-selective halogenation method for pyridines, quinolines, and isoquinolines via electrophilic halogen radical addition utilizing an N-benzyl activation strategy. This method achieves C3- and C5-dihalogenation in pyridines, C3- and C6-dihalogenation in quinolines, and C3-monohalogenation in isoquinolines. The feasibility and potential applications of this method were validated through scale-up reactions and the bromination of quinoline derivatives with biomolecular fragments.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


