通过使用改良施密特型反应的探索实验在重排反应中引入区域选择性
IF 2.5
3区 教育学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
重排反应是有机化学中最重要的转化之一,是该学科的一个重要而又困难的方面。然而,传统的实验教科书中很少涉及涉及区域选择性的重排反应。在此,我们设计并展示了一个以施密特型重排反应为中心的新型有机化学实验,旨在为学生阐明该反应的机理及其区域选择性。在进行实验之前,先让学生预习 4-硝基二苯甲酮与 n-PrNO2 反应生成酰胺产物的机理。鉴于酮的不对称性质,获得不同酰胺产物的可能性取决于 4-硝基二苯甲酮的两个芳基在重排过程中迁移的优先顺序。通过本实验,学生可以发现产物产生于电子更丰富的芳基的迁移,从而表明亲核重排具有明显的区域选择性。此外,绿色化学的概念已融入有机化学课程,强调了这一改进的施密特型反应在减少毒性和提高安全性方面的贡献。这种方法促使学生认识到绿色化学在推动社会可持续发展方面的必要性,强调化学家肩负环保意识的集体责任。本文章由计算机程序翻译,如有差异,请以英文原文为准。
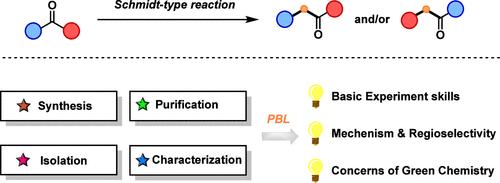
Introducing Regioselectivity in Rearrangement Reactions through a Discovery Experiment Using an Improved Schmidt-Type Reaction
Rearrangement reactions, one of the most important transformations in organic chemistry, represent an important yet difficult aspect of the discipline. However, rearrangement reactions that involve regioselectivity are rarely covered in traditional experimental textbooks. Herein, a novel organic chemistry laboratory experiment centered around a Schmidt-type rearrangement was designed and presented, aimed at elucidating the mechanism of this reaction and its regioselective nature for students. Before conducting the experiment, students are tasked with previewing the mechanism underlying the formation of the observed amide product from the reaction between 4-nitrobenzophenone and n-PrNO2. Given the asymmetric nature of the ketone, the potential for obtaining different amide products depends on the priority of the migration of the two aryls of 4-nitrobenzophenone during rearrangement. Through this experiment, students can discover that the product arises from the migration of the more electron-rich aryl, thereby indicating a nucleophilic rearrangement with evident regioselectivity. Additionally, the concept of green chemistry was integrated into the organic chemistry curriculum, underscoring the contribution of this improved Schmidt-type reaction in reducing toxicity and enhancing safety. This approach prompts students to recognize the imperative of green chemistry in advancing sustainable societal development, emphasizing the collective responsibility of chemists to shoulder environmentally conscious practices.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Chemical Education
化学-化学综合
CiteScore
5.60
自引率
50.00%
发文量
465
审稿时长
6.5 months
期刊介绍:
The Journal of Chemical Education is the official journal of the Division of Chemical Education of the American Chemical Society, co-published with the American Chemical Society Publications Division. Launched in 1924, the Journal of Chemical Education is the world’s premier chemical education journal. The Journal publishes peer-reviewed articles and related information as a resource to those in the field of chemical education and to those institutions that serve them. JCE typically addresses chemical content, activities, laboratory experiments, instructional methods, and pedagogies. The Journal serves as a means of communication among people across the world who are interested in the teaching and learning of chemistry. This includes instructors of chemistry from middle school through graduate school, professional staff who support these teaching activities, as well as some scientists in commerce, industry, and government.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


