在可见光照射下催化还原三氟甲基化烷基溴和合成烷基杂环:卤素键和镍催化作用的协同作用
IF 4.7
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
我们探索了在可见光照射下卤素键与镍催化之间的协同作用。该反应体系可在一个反应锅中同时还原α-三氟甲基溴化烷基,以及活化四氢呋喃(THF)与杂环的交叉偶联。镍催化剂和可见光活化的战略整合,再加上卤素键的辅助作用,使转化过程具有显著的原子经济性,并表现出优异的官能团耐受性。通过对具有生物活性的分子进行直接修饰,这一方案的实用性得到了进一步强调。本文章由计算机程序翻译,如有差异,请以英文原文为准。
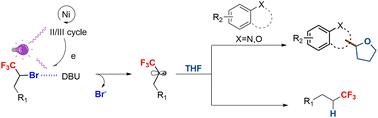
Catalytic reduction of trifluoromethylated alkyl bromides and synthesis of alkylated heterocycles under visible light irradiation: synergetic action of a halogen bond and Ni catalysis†
The synergistic interplay between a halogen bond and Ni catalysis under visible light irradiation has been explored. This reaction system enables the concurrent reduction of α-trifluoromethyl alkyl bromides and the cross-coupling of activated tetrahydrofuran (THF) with heterocycles in one pot. This strategic integration of nickel catalysts and visible light activation, complemented by the auxiliary effect of the halogen bond, renders the transformation remarkably atom-economic and demonstrates exceptional functional group tolerance. The practical utility of this protocol has been further underscored through the direct modification of biologically active molecules.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic Chemistry Frontiers
CHEMISTRY, ORGANIC-
CiteScore
7.90
自引率
11.10%
发文量
686
审稿时长
1 months
期刊介绍:
Organic Chemistry Frontiers is an esteemed journal that publishes high-quality research across the field of organic chemistry. It places a significant emphasis on studies that contribute substantially to the field by introducing new or significantly improved protocols and methodologies. The journal covers a wide array of topics which include, but are not limited to, organic synthesis, the development of synthetic methodologies, catalysis, natural products, functional organic materials, supramolecular and macromolecular chemistry, as well as physical and computational organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


