导出素 1 通过 ERK1/2 核导出调节肿瘤中髓源性抑制细胞的免疫抑制功能。
IF 21.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
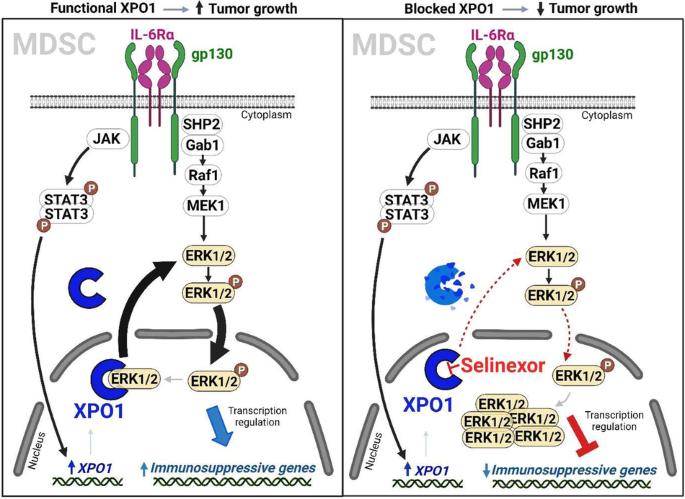
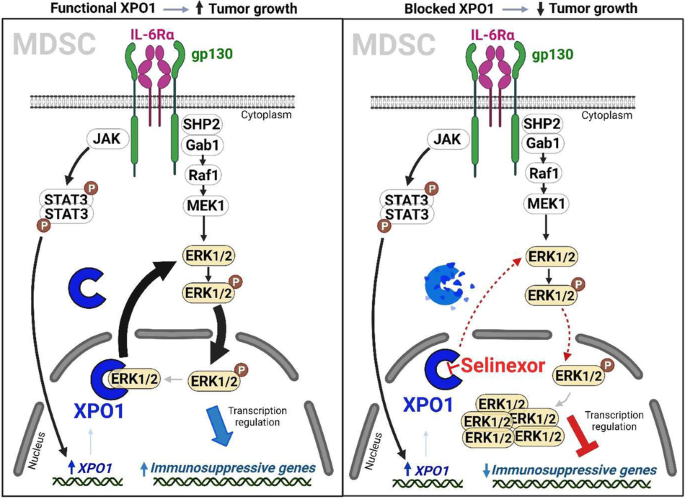
髓源性抑制细胞(MDSCs)是肿瘤免疫抑制的主要驱动因素。了解决定这些细胞的发育和免疫抑制功能的机制可以为改善抗肿瘤免疫提供新的治疗靶点。在这里,我们利用临床前小鼠模型发现,肿瘤 MDSCs 中的 exportin 1(XPO1)表达上调,而这种上调是由 MDSC 分化过程中 IL-6 诱导的 STAT3 激活诱导的。阻断 XPO1 可将 MDSCs 转化为激活 T 细胞的中性粒细胞样细胞,从而增强抗肿瘤免疫反应并抑制肿瘤生长。从机理上讲,XPO1抑制会导致ERK1/2的核禁锢,从而阻止IL-6介导的MAPK信号通路激活后的ERK1/2磷酸化。同样,在人类 MDSCs 中阻断 XPO1 可诱导形成具有免疫刺激功能的中性粒细胞样细胞。因此,我们的研究结果揭示了XPO1在MDSC分化和抑制功能中的关键作用;利用这些新发现揭示了重编程免疫抑制MDSC以改善癌症治疗反应的新靶点。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Exportin 1 governs the immunosuppressive functions of myeloid-derived suppressor cells in tumors through ERK1/2 nuclear export
Myeloid-derived suppressor cells (MDSCs) are a main driver of immunosuppression in tumors. Understanding the mechanisms that determine the development and immunosuppressive function of these cells could provide new therapeutic targets to improve antitumor immunity. Here, using preclinical murine models, we discovered that exportin 1 (XPO1) expression is upregulated in tumor MDSCs and that this upregulation is induced by IL-6-induced STAT3 activation during MDSC differentiation. XPO1 blockade transforms MDSCs into T-cell-activating neutrophil-like cells, enhancing the antitumor immune response and restraining tumor growth. Mechanistically, XPO1 inhibition leads to the nuclear entrapment of ERK1/2, resulting in the prevention of ERK1/2 phosphorylation following the IL-6-mediated activation of the MAPK signaling pathway. Similarly, XPO1 blockade in human MDSCs induces the formation of neutrophil-like cells with immunostimulatory functions. Therefore, our findings revealed a critical role for XPO1 in MDSC differentiation and suppressive functions; exploiting these new discoveries revealed new targets for reprogramming immunosuppressive MDSCs to improve cancer therapeutic responses.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


