使用生物活化的体内组装肽清除嗜纤维化巨噬细胞可改善肾脏纤维化。
IF 21.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
由于患病肾脏中存在复杂的细胞信号冗余,治疗肾脏纤维化具有挑战性。肾脏纤维化涉及以巨噬细胞为主的免疫反应,巨噬细胞会激活纤维化龛位中的肌成纤维细胞。然而,巨噬细胞表现出高度异质性,阻碍了其作为治疗细胞靶点的潜力。在此,我们旨在从时间和空间上消除驱动肾脏廓清性免疫反应的特定巨噬细胞亚群。我们确定了肾脏中主要的凋亡性巨噬细胞亚群(Fn1+Spp1+Arg1+),然后构建了一种被命名为生物活化体内组装 PK(BIVA-PK)的 12-mer糖肽来清除这些细胞。BIVA-PK 能特异性地与坏死性巨噬细胞结合并被其内化。通过诱导巨噬细胞死亡,BIVA-PK重塑了肾脏微环境并抑制了廓清性免疫反应。BIVA-PK在改善肾脏纤维化和保护肾功能方面的强大疗效凸显了以巨噬细胞亚群为靶点作为慢性肾脏病患者潜在疗法的价值。本文章由计算机程序翻译,如有差异,请以英文原文为准。
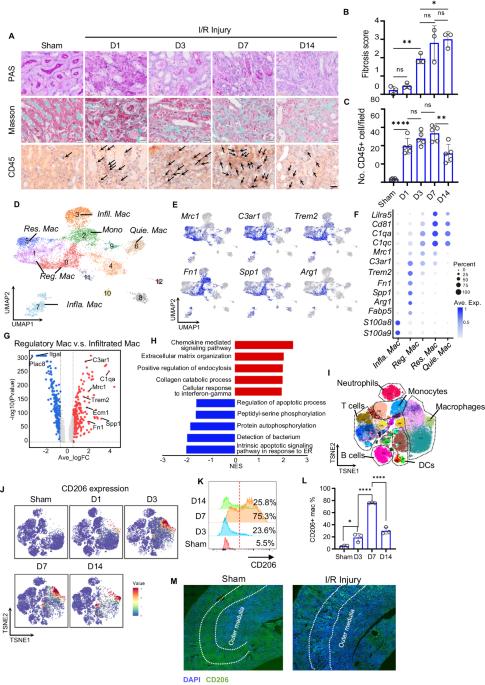
Depleting profibrotic macrophages using bioactivated in vivo assembly peptides ameliorates kidney fibrosis
Managing renal fibrosis is challenging owing to the complex cell signaling redundancy in diseased kidneys. Renal fibrosis involves an immune response dominated by macrophages, which activates myofibroblasts in fibrotic niches. However, macrophages exhibit high heterogeneity, hindering their potential as therapeutic cell targets. Herein, we aimed to eliminate specific macrophage subsets that drive the profibrotic immune response in the kidney both temporally and spatially. We identified the major profibrotic macrophage subset (Fn1+Spp1+Arg1+) in the kidney and then constructed a 12-mer glycopeptide that was designated as bioactivated in vivo assembly PK (BIVA-PK) to deplete these cells. BIVA-PK specifically binds to and is internalized by profibrotic macrophages. By inducing macrophage cell death, BIVA-PK reshaped the renal microenvironment and suppressed profibrotic immune responses. The robust efficacy of BIVA-PK in ameliorating renal fibrosis and preserving kidney function highlights the value of targeting macrophage subsets as a potential therapy for patients with CKD.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


